Take-home points
|
 |
Bios Dr. London is president of the San Diego Retina Research Foundation, the founder and president of the Ophthalmology Society of San Diego and chief of Ophthalmology at Scripps Memorial Hospital in La Jolla, Calif. Disclosures: The authors have no relevant financial disclosures. |
Anti-VEGF therapy transformed neovascular AMD from a blinding disease to one we can manage. But we still argue about the endpoint: complete anatomic dryness on OCT, or tolerance of some residual fluid, particularly small-volume subretinal fluid. My practice reflects the evidence through 2025, which consistently favors treating until dry: completely eliminating intraretinal fluid and minimizing subretinal fluid to optimize long-term vision.
Not all fluid behaves the same
Post-hoc analyses of CATT, HARBOR, and VIEW taught us that intraretinal fluid, especially when it involves the fovea, predicts worse visual outcomes.1,10 In HARBOR, residual IRF meant diminished vision gains regardless of location or severity.1 Subretinal fluid showed a more variable relationship with acuity, and residual SRF at 24 months didn’t correlate with worse vision in that analysis.
But we shouldn’t oversimplify this into blanket SRF tolerance. A 2025 systematic review found persistent fluid in 41 percent of eyes at one year and 47 percent at two years despite ongoing treatment.3 The key finding: Mean visual acuity was significantly better in eyes without any fluid. Limited SRF may be tolerable in select cases under rigorous follow-up and monitoring protocols, but across broad patient populations in the real world, a completely dry retina consistently delivers superior functional outcomes.
What FLUID actually tells us
The FLUID study gets cited frequently to justify fluid tolerance. It demonstrated non-inferior vision when allowing up to 200 µm of SRF in a treat-and-extend protocol.2 Look closer at what FLUID required: continuous anti-VEGF therapy regardless of fluid status—treatment never stopped. IRF resolution was mandatory before extending intervals. And the injection burden difference was modest, only 1.2 fewer injections over two years (15.8 versus 17).
FLUID supports selective SRF tolerance only within rigorous, ongoing therapy with mandatory IRF clearance. It’s not a license for undertreatment or accepting any IRF.
The real-world problem: We undertreat
Clinical trials run on idealized schedules with protocol-driven imaging and strict retreatment criteria. Real-world registries tell a different story: fewer injections, longer intervals, and delayed detection of recurrence.5,6 These patterns consistently produce worse visual outcomes than trial results.
When you adopt a fluid-tolerant strategy in real-world practice, you risk normalizing undertreatment. The TRUCKEE study illustrates this perfectly.7 Among patients switched to faricimab because of suboptimal response to prior therapy, more than 80 percent still had residual IRF and more than 60 percent had residual SRF after the first injection. These numbers highlight both how common persistent fluid is in routine practice and how difficult achieving dryness can be when patients have been undertreated.
Fluid fluctuations cause particular harm. Recent HAWK and HARRIER analyses as well as real-world data confirm that greater fluid stability—more visits with dry retinas—correlates with larger vision gains.9, 10 In fact, patients whose retinas were consistently dry after loading had better visual and anatomic outcomes.11 SRF that fluctuates or increases between visits often heralds IRF recurrence and progressive photoreceptor injury. That small amount of SRF you note at one visit can become significant by the next if you’ve stretched the interval or if imaging review isn’t meticulous.
OCT interpretation isn’t as reliable as we think
Here’s something we don’t discuss enough: OCT interpretation is operator-dependent. CATT data showed treating ophthalmologists miss or misclassify fluid compared with reading centers.8 This matters enormously when you adopt permissive strategies, you’re more likely to miss opportunities for timely retreatment.
Treating to anatomic dryness gives you a clear, objective endpoint, as well as a clear indication of increased or recurrent disease activity. It eliminates the interpretive ambiguity about what constitutes “acceptable” residual fluid and reduces the risk of silent progression between visits.
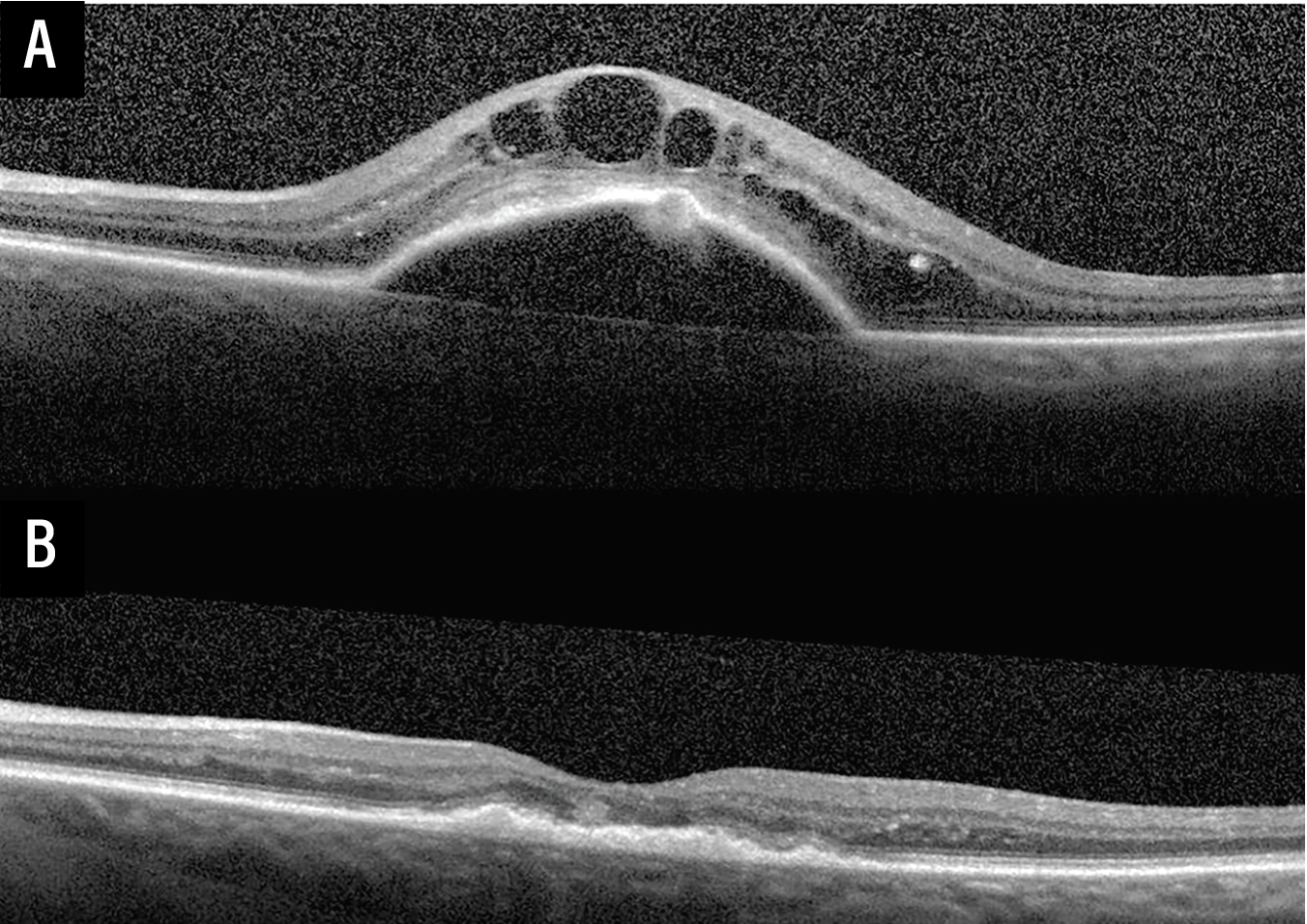 |
| Figure 1. Optical coherence tomography images demonstrating treatment response to intravitreal anti-VEGF therapy in treatment naive neovascular age-related macular degeneration. (A) Baseline OCT of the left eye at the time of presentation with neovascular AMD demonstrating a fibrovascular pigment epithelial detachment, subretinal hyperreflective material (SRHM), and intraretinal fluid. (B) Follow-up OCT of the same eye following three monthly loading intravitreal anti-VEGF injections demonstrating marked reduction in PED height and complete resolution of IRF—illustrating the robust anatomic response that supports a treat-until-dry management approach. |
Long-term structural damage from persistent fluid
Chronic fluid exacts a structural toll beyond immediate acuity measures. Real-world data link persistent fluid to outer retinal atrophy, complete RPE and outer retinal atrophy, and increased subretinal fibrosis, all strongly tied to visual decline. About half of nAMD patients develop subretinal fibrosis by two years despite anti-VEGF therapy. Fibrosis and atrophy together drive irreversible vision loss.
The ellipsoid zone serves as our key biomarker for photoreceptor health. Chronic IRF correlates with progressive EZ disruption and consequent vision loss. Each fluid recurrence likely inflicts incremental photoreceptor damage that accumulates over years. Early and sustained fluid resolution preserves the retinal microarchitecture photoreceptors needed for long-term function.
How I implement this approach
I use treat-and-extend with a low threshold for treating new or persistent fluid. After three monthly loading doses, I extend intervals only when IRF is fully resolved and SRF is minimal or absent. High-quality OCT matters. I review the entire macular volume, not just central subfield thickness, looking carefully for subtle parafoveal IRF and assessing EZ integrity. This approach allows me to strike a balance between the known risks of undertreatment, while also avoiding potential risks associated with ongoing monthly therapy.
Patient education on adherence is critical. The “treat until dry” philosophy only works with sustained engagement over years of care.
Balancing aggressive treatment with individualization
My goal is anatomic quiescence—a dry retina—that permits safe interval extension. Many patients reach 12-week intervals while maintaining complete fluid resolution; others need more frequent treatment. I guide management by anatomic endpoints, not arbitrary calendar-based intervals, which allows for appropriately tailored therapy in a population of patients with heterogeneous medication-response and underlying disease activity.
Clinical judgment remains essential. For frail or elderly patients where treatment risks outweigh benefits or goals differ, I modify the approach after careful discussion. But for most nAMD patients, aggressive fluid elimination offers the best chance at preserving vision long-term. With patients living longer and healthier than ever before, this is what matters to them. Emerging agents and delivery systems (higher-dose aflibercept, brolucizumab, Susvimo, gene therapies like sura-vec/ABBV-RGX-314) aim to maintain dryness with fewer interventions, which strengthens this philosophy.
The atrophy question
The concern about intensive anti-VEGF dosing accelerating geographic atrophy is legitimate. CATT showed higher GA risk with monthly versus PRN dosing,4 and VEGF is a trophic factor for RPE, photoreceptors and choriocapillaris.
But untreated or inadequately treated CNV, variation in retinal thickness and chronic fluid also drive atrophy through inflammation, RPE dysfunction and photoreceptor loss. The question becomes: What causes more harm, recurrent inadequately controlled exudation or VEGF suppression itself? Current evidence supports achieving early stability (a dry retina), then carefully extending intervals to minimize both cumulative anti-VEGF exposure and tissue damage from recurrent fluid.
Conclusion
Fluid-tolerance data have refined our approach, but the bulk of evidence through 2025 supports treating nAMD until the retina is dry. This strategy optimizes visual acuity, preserves critical microarchitecture and reduces long-term complications like fibrosis and atrophy. In the real world, where follow-up and imaging quality vary, an explicit treat-to-dry strategy reduces undertreatment risk and provides a clear therapeutic target.
“Treat until dry” isn’t overtreatment—it’s adequate treatment. That distinction makes all the difference in preserving our patients’ vision over years of disease. RS
REFERENCES
1. Holekamp NM, et al. Relationship between retinal fluid characteristics and vision in neovascular age-related macular degeneration: HARBOR post hoc analysis. Br J Ophthalmol 2022;106:11:1595-1603.
2. Guymer RH, et al. Tolerating subretinal fluid in neovascular age-related macular degeneration treated with ranibizumab using a treat-and-extend regimen: FLUID study 24-month results. Ophthalmology 2019;126:5:723-734.
3. Patil NS, et al. Persistence of retinal fluid after anti-VEGF treatment for neovascular age-related macular degeneration: A systematic review and meta-analysis. Ophthalmol Retina 2025;S2468-6530:25:00012-0.
4. Grunwald JE, et al. Risk of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology 2014;121:1:150-161.
5. Wykoff CC, et al. Impact of anti-VEGF treatment and patient characteristics on vision outcomes in neovascular AMD: Up to 6-year analysis of the AAO IRIS Registry. Ophthalmol Sci 2023;4:2:100421.
6. Holz FG, et al. Key drivers of visual acuity gains in neovascular AMD in real life: Findings from the AURA study. Br J Ophthalmol 2016;100:12:1623-8.
7. Khanani AM, et al. The real-world efficacy and safety of faricimab in neovascular age-related macular degeneration: The TRUCKEE study – 6 month results. Eye 2023;37:3574-81.
8. Toth CA, et al. Identification of fluid on optical coherence tomography by treating ophthalmologists versus a reading center in the CATT. Retina 2015;35:7:1303-14.
9. Chakravarthy U, et al. Impact of macular fluid volume fluctuations on visual acuity during anti-VEGF therapy in eyes with nAMD. Eye (Lond) 2021;35:11:2983-90.
10. Dugel PU, et al. Effect of retinal thickness variability on visual outcomes and fluid persistence in neovascular age-related macular degeneration: a post hoc analysis of HAWK and HARRIER studies. Retina 2022;42:3:511-518.
11. Eichenbaum, et al. Impact of retinal fluid-free months on outcomes in neovascular age-related macular degeneration: a treatment agnostic analysis of the HAWK and HARRIER studies. Retina 2023;43:4:632-640.