Take-home points
|
 |
Bios Dr. Guymer is deputy director of the Centre for Eye Research Australia, the head of macular research at CERA, and professor of ophthalmology at the University of Melbourne. She's also a senior retinal specialist at The Royal Victorian Eye and Ear Hospital. Disclosures: The authors have no relevant financial disclosures. |
As we moved from a “one size fits all” anti-vascular endothelial growth factor treatment for exudative macular neovascularization, secondary to age-related macular degeneration, to individualized treatment intervals, it was necessary for clinicians to make an assessment of the VEGF-driven disease activity. In the now widely adopted treat-and-extend regimens for anti-VEGF treatment in neovascular AMD, determining VEGF-driven disease activity is required, as it dictates the next treatment interval: increasing the interval if the disease is deemed inactive, or decreasing it if the disease is active. Identifying ongoing disease activity in the setting of ongoing treatment for nAMD largely comes down to the presence or absence of fluid in the retina. Thus, the presence of black spaces on an OCT scan has become the surrogate biomarker for VEGF-driven disease activity. Unfortunately, this biomarker isn’t perfect, its presence doesn’t always imply VEGF-driven exudative neovascular disease activity, and as such requires a nuanced interpretation, both at diagnosis and in the ongoing management of nAMD.1
At diagnosis, clinicians need to be sure the black spaces in the retina, which may at times be discovered incidentally on routine imaging, without any symptoms, are truly due to exudative nAMD, and not another cause commonly misinterpreted as exudative AMD such as pseudovitelliform lesions, central serous chorioretinopathy, macular telangiectasis (MacTel), draping between two drusenoid PEDs, or fluid not uncommonly seen at the apex of large pigment epithelial detachments (PEDs) or degenerative cysts associated with the early signs of atrophy.2-5
Assuming the correct diagnosis, then treatment-naïve exudative MNV should be treated without delay, with the initial “loading” phase of treatment aiming to stop the exudation as rapidly as possible. The aim is to return the retinal anatomy to as near normal as possible, as soon as possible.
The question is: What to do if, despite this intense initial phase of treatment (or at some later time point in treatment) and despite ongoing regular treatment every four to six weeks, not all fluid is resolved? In a strict treat-and-extend protocol, which allows for no fluid before interval extension, the intervals shouldn’t be increased. This would then mean that a patient might be treated monthly with an anti-VEGF indefinitely if the fluid fails to completely resolve, and the eye will be labeled a “hungry” eye or a “failure to respond” eye. Likely the patient will be changed to different drugs in the hope of extending the interval between injections, often with no success. This patient might even be enrolled into a new drug trial for people who are deemed to “require” frequent injections.
Is there another way to handle this scenario? Clearly if the fluid is increasing, or new, one has to assume that it’s due to VEGF-driven diseases (even if there is some doubt), but because it’s new/increasing, the safest option (first do no harm) is to assume it’s due to VEGF and decrease the interval to see if you can improve the situation.
The question is really around stable fluid—give or take a few microns. Could some apparently stable fluid be tolerated, allowing the interval to increase and the treatment burden to be reduced?
Dealing with intraretinal fluid first. Exudative cysts are defined by their quick resolution after anti-VEGF therapy, whereas degenerative cysts persist.6 Many studies have shown that IRF, secondary to exudative nAMD, is associated with poorer visual acuity outcomes and an increased risk of developing atrophy and fibrosis, both of which contribute to vision loss.7-10 In the CATT trial, with respect to fluid, IRF was found to have the greatest impact on vision at all time points.11
Thus, IRF should be treated, with the aim to resolve these cysts, but if they persist, then consider if they could be atrophic. This might especially be the case if they appear above areas suggestive of early atrophy such as signal hypertransmission into the choroid, or retinal pigment epithelial loss.12
Also consider: Could the cystic spaces not be a result of nAMD? This is where fluorescein angiography would help ensure the diagnosis is correct, proving that these spaces aren’t due to diseases such as MacTel. In the setting of nAMD, if IRF isn’t going away or decreasing, despite intensive treatment with short intervals, consider cautious extension, even by one week at a time. If the IRF increases, then you have the answer—they’re likely a result of VEGF-driven disease, so they appear to need ongoing short-interval treatment. If they stay the same, then try another week’s extension.
With regard to tolerating fluid in the setting of true exudative nAMD, the main question to consider relates to subretinal fluid. One scenario to explain stable SRF not responding to anti-VEGF, in the setting of actual nAMD, is that these black spaces aren’t related to ongoing exudation and unchecked VEGF activity, but rather may be a result of draping, failure of the outer retina to reattach to the RPE, possibly due to osmotic pressure in the space due to residual material. In which case, one could consider slowly extending the interval, perhaps by only a week at a time. If, in so doing, the fluid increases, then one has to assume it’s active, exudative, VEGF-driven disease, but if it stays relatively the same then continuing cautious extension should be considered.
Another scenario is that possibly the persistence of SRF in the setting of nAMD is truly indicative of a small amount of exudation. But maybe this isn’t all bad. The presence of SRF at baseline and persistence of SRF have been associated with better long-term visual acuity.7-9
Thus, there’s evidence to suggest that allowing some residual SRF isn’t detrimental and may actually be beneficial to vision. In an analysis of data from patients with nAMD in the IVAN trial, the presence of SRF at the final visit halved the probability of atrophy developing within the nAMD lesion (odds ratio [OR] 0.41; p=0.004),13 with similar findings reported in other studies.14,15
One study in nAMD found that the presence of SRF at 36 months was associated with a lower rate of fibrosis (OR 0.49; 95% confidence interval [CI] 0.29–0.81) and fibrosis progression (OR 0.50; 95% CI 0.31–0.81) (both p=0.005).15 In these studies, poor visual outcomes are often associated with the development of atrophy and fibrosis rather than with incomplete treatment of VEGF-driven MNV activity.16
It’s possible that the maturing MNV under the RPE “oozes” a little from time to time but does no harm, and indeed is providing oxygen and nutrients to reduce the risk of atrophy.
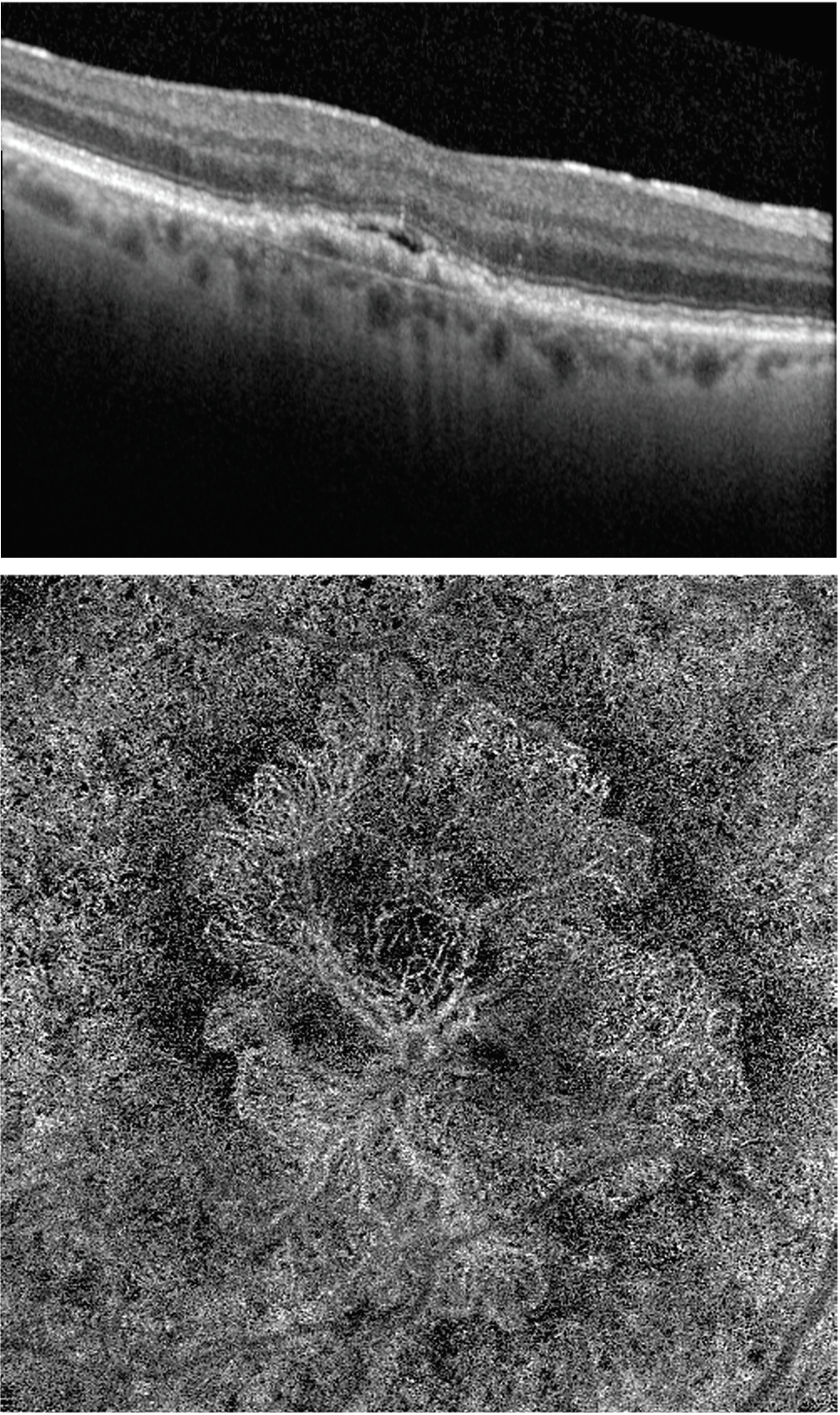 |
| Figure 1. OCT B Scan of a patient who’d been treated with anti-VEGF treatment two years ago. A small amount of SRF persists while visual acuity remains stable and OCT angiography shows the large mature vessel complex. |
Even under prolonged VEGF suppression, persistence of flow within mature vessels has been demonstrated in eyes with type 1 neovascular lesions (Figure 1).16
In these cases of stable SRF, tolerating a small amount of SRF and small fluctuations in the SRF may be okay, with the possibility that the ultimate aim of treatment is to achieve a stage of mature non-exudative vessels that continue to nourish the outer retina, but from time to time may leak a little.
The FLUID study was a pioneering work, advocating a tolerant approach towards small amounts of residual SRF.17 Recent American Society of Retina Specialists surveys tried to gauge physicians’ tolerance for fluid in nAMD, and found that consistently around half of participating physicians indicated that they would maintain treatment intervals and tolerate some SRF.18
Yet clinical practice guidelines from the American Academy of Ophthalmology, the Royal College of Ophthalmologists in the United Kingdom and the European Society of Retina Specialists all state that fluid on OCT is an indication of active disease and recommend retreatment when fluid is present.19-21
However, the Vision Academy recently published a consensus guidelines paper where they state that “small amounts of persistent fluid could be tolerated without compromising vision.”22
Satisfactory outcomes can be achieved by cautious tolerance of some residual, stable SRF. This approach allows patients to be treated using fewer injections, as suggested in the FLUID trial, where even small increases in the interval make a large difference over the patient’s long-term treatment and to the health-care burden. RS
REFERENCES
1. Lek JJ, Caruso E, Baglin EK, et al. Interpretation of subretinal fluid using OCT in intermediate age-related macular degeneration. Ophthalmol Retina 2018;2:792–802.
2. Hilely A, Au A, Freund KB, et al. Non-neovascular age-related macular degeneration with subretinal fluid. Br J Ophthalmol 2021;105:1415–20
3. Madjarov B, Rosenfeld PJ. Serous PEDs with associated subretinal fluid unresponsive to anti-VEGF therapy. Invest Ophthalmol Vis Sci 2009;50:238
4. Sikorski BL, Bukowska D, Kaluzny JJ, Szkulmo-wski M, Kowalczyk A, Wojtkowski M. Drusen with accompanying fluid underneath the sensory retina. Ophthalmology 2011;118:82–92.
5. Adrean SD, Chaili S, Pirouz A, Grant S. Central serous chorioretinopathy in elderly patients mimicking occult neovascular age-related macular degeneration. Clin Ophthalmol 2020;14:4073–8.
6. Arrigo A, Aragona E, Battaglia Parodi M, Bandello F. Quantitative multimodal imaging characterization of intraretinal cysts versus degenerative pseudocysts in neovascular age-related macular degeneration. Ophthalmol Retina 2024;8:1118.
7. Ritter M, Simader C, Bolz M, et al. Intraretinal cysts are the most relevant prognostic biomarker in neovascular age-related macular degeneration independent of the therapeutic strategy. Br J Ophthalmol 2014;98:1629–35.
8. Waldstein SM, Wright J, Warburton J, Margaron P, Simader C, Schmidt-Erfurth U. Predictive value of retinal morphology for visual acuity outcomes of different ranibizumab treatment regimens for neovascular AMD. Ophthalmology 2016;123:60–9.
9. Waldstein SM, Simader C, Staurenghi G, et al. Morphology and visual acuity in aflibercept and ranibizumab therapy for neovascular age-related macular degeneration in the VIEW trials. Ophthalmology 2016;123:1521–9.
10. Sadda SR, Tuomi LL, Ding B, Fung AE, Hopkins JJ. Macular atrophy in the HARBOR study for neo-ascular age-related macular degeneration. Ophthalmology 2018;125:878–86.
11. Jaffe GJ, Ying GS, Toth CA, et al. Macular morphology and visual acuity in year five of the comparison of age-related macular degeneration treatments trials. Ophthalmology 2019;126:252–60.
12. Guymer RH, Rosenfeld PJ, Curcio CA, et al. Incomplete retinal pigment epithelial and outer retinal atrophy in age-related macular degeneration: Classification of Atrophy Meeting Report 4. Ophthalmology 2020;127:394-409.
13. Bailey C, Scott LJ, Rogers CA, et al. Intralesional macular atrophy in anti-vascular endothelial growth factor therapy for age-related macular degeneration in the IVAN trial. Ophthalmology 2019;126:75–86.
14. Sánchez-Monroy J, Nguyen V, Puzo M, et al. Sub-retinal fluid may protect against macular atrophy in neovascular age-related macular degeneration: 5 years of follow-up from Fight Retinal Blindness registry. Acta Ophthalmol 2023;101:457–64.
15. Llorente-González S, Hernandez M, González-Zamora J, et al. The role of retinal fluid location in atrophy and fibrosis evolution of patients with neovascular age-related macular degeneration long-term treated in real world. Acta Ophthalmol 2022;100:e521–31.
16. Rofagha S, Bhisitkul RB, Boyer DS, et al. Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: A multicenter cohort study (SEVEN-UP). Ophthalmology 2013;120:2292–99.
17. Guymer RH, Markey CM, McAllister IL, et al. Tolerating subretinal fluid in neovascular age-related macular degeneration treated with ranibizumab using a treat-and-extend regimen: FLUID study 24-month results. Ophthalmology 2019;126:723–34.
18. Hahn P, Eliott D, eds. 2021 Global Trends in Retina Survey [online]. 2021. https://www.asrs.org/content/documents/2021-global-trends-survey-highlights.pdf
19. Schmidt-Erfurth U, Chong V, Loewenstein A, et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol 2014;98:1144–67.
20. Flaxel CJ, Adelman RA, Bailey ST, et al. Age-Related Macular Degeneration Preferred Practice Pattern. Ophthalmology 2020;127:PP1–P65.
21. Chakravarthy U, Williams M, AMD Guidelines Group. The Royal College of Ophthalmologists Guidelines on AMD: Executive Summary. Eye 2013;27:1429–31.
22. Zur D, Guymer R, Korobelnik JF, Wu L, Viola F, Eter N, Baillif S, Chen Y, Arnold JJ. Impact of residual retinal fluid on treatment outcomes in neovascular age-related macular degeneration. British Journal of Ophthalmology 2025;109:3:307-15.