|
Bios |
Diabetic tractional retinal detachment cases are daunting and unpredictable for any vitreoretinal surgeon. The primary concern during surgery is the ability to relieve all tractional components while avoiding the creation of an iatrogenic retinal break. A retinal break increases surgical complexity, often requiring limited retinectomy, silicone oil tamponade, or scleral buckle to relieve traction and enable endolaser uptake.1 Postoperatively, the risk of proliferative vitreoretinopathy rises, and posterior breaks may lead to poor visual outcomes.2
Technological advances have enabled the development and growing popularity of small-gauge vitrectomy, such as the 27-ga. vitrectomy described by Osaka’s Yusuke Oshima, MD, PhD, in 2010.3 27-gauge vitrectomy has been shown to result in minimal complications and favorable postoperative visual outcomes.4 Here, we describe a 27-ga. single-gauge pars plana vitrectomy approach for a young diabetic patient with aggressive “wolf-jaw” configuration5 TRD and vitreous hemorrhage.
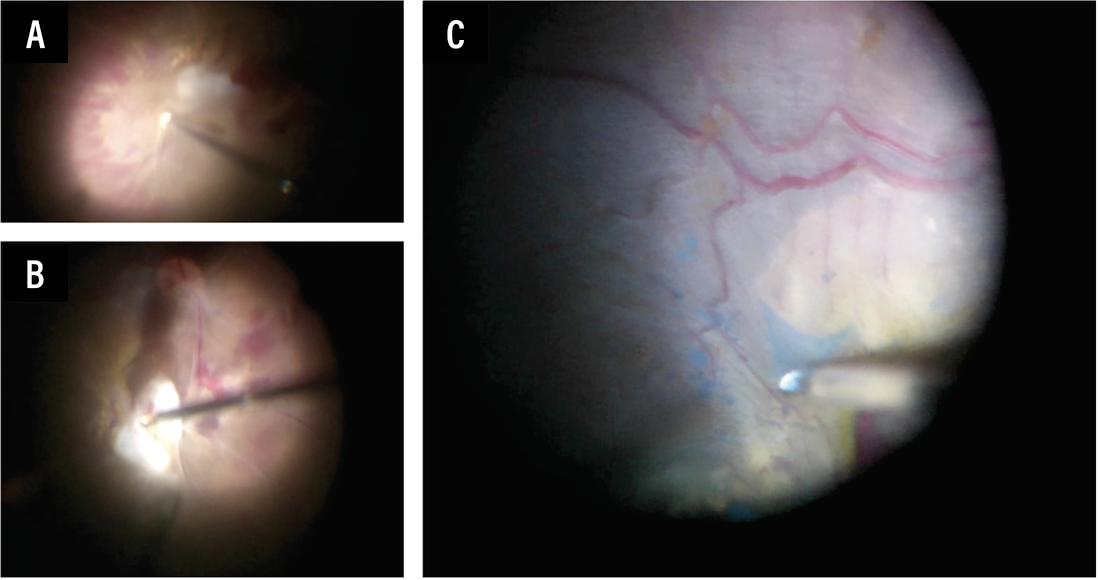 |
| Figure 1. A) Navigating tight surgical spaces with the 27-gauge vitrector to release fibrovascular traction and delamination further with proportion reflux hydro-dissection. B) Using end-gripping forceps to exert gentle tension on the membranes to release any underlying fibrovascular pegs. C) ILM peel is performed over the macula to relieve any residual traction. |
The surgical “grazing” technique
In this case, the tractional bands were extensive over the posterior pole and the macula. An intravitreal injection of anti-vascular endothelial growth factor was administered three days preoperatively to improve intraoperative safety. A single 27-gauge system was used with a high-speed 10K vitrectomy cutter (Figure 1). Given the patient’s young age, we didn’t perform a lensectomy. With a concurrent bleached vitreous hemorrhage, core vitrectomy was performed with caution. We find that positioning the cutter facing upwards in the mid-vitreous cavity is relatively safe until better visualization is gained. The posterior hyaloid is then identified and entered to create a surgical plane to relieve the tractional forces on the posterior pole.
Once the posterior pole is isolated, persistence is key in delaminating and segmenting the fibrovascular membrane without causing a break. We used the vitrector with reduced vacuum settings, ensuring the bevel was strictly away from the retina, entering tight retinal spaces to “graze” on the fibrovascular membranes. End-gripping (such as internal limiting membrane) forceps were used when necessary. When using forceps, gentle tension should be applied; if the retina appears too taut, further delamination may be required. The 27-gauge vitrector allows for navigation through tight surgical spaces, and proportional reflux hydrodissection is used as an adjunct to create a fluid-based tissue separation. It’s acceptable to leave small, isolated residual fibrovascular pegs as long as all traction is alleviated. ILM peeling is advised when significant macular membrane or distortion is present to further relieve traction and reduce the risk of membrane re-proliferation over the macula.
Completion of panretinal endolaser is essential to reduce ischemic drive postoperatively. In the absence of retinal breaks and with adequate delamination, fluid-air exchange can be performed, leaving only air tamponade in situ.
Color fundus preoperative and postoperative images are illustrated in Figure 2.
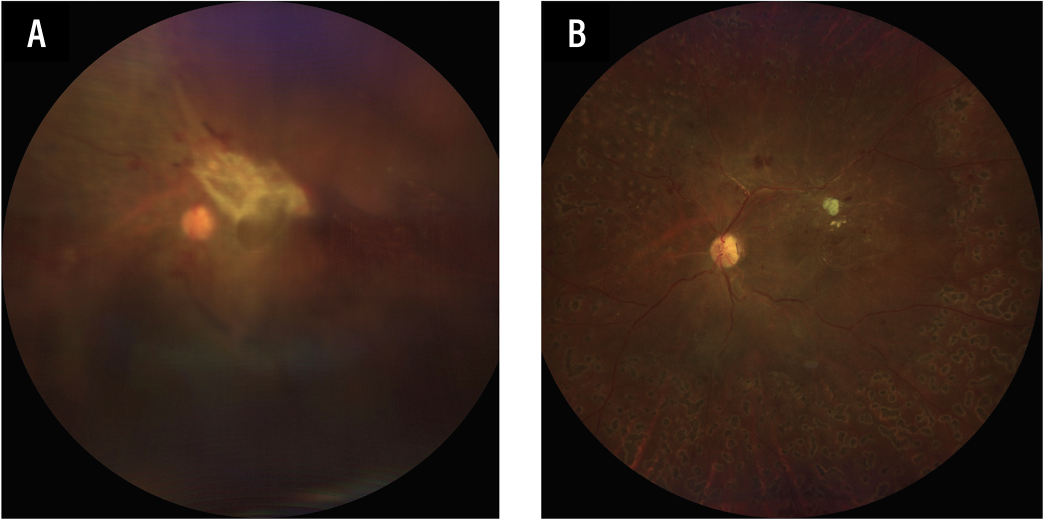 |
| Figure 2. A) Initial preoperative fundus photograph showing central dispersed vitreous hemorrhage, with “wolf-jaw” configuration TRD. B) Postoperative fundus photograph showing reattached retina with isolated residual fibrovascular stump. |
The bottom line
With meticulous and purposeful surgical maneuvers, a single 27-gauge vitrectomy allows for precise segmentation and delamination while navigating through tight surgical planes and adherent fibrovascular bands. This approach minimizes the risk of iatrogenic retinal breaks, negates the need for additional chandelier placement, reduces surgical complexity and surgical time, and maintains a good anatomical outcome. RS
References
1. Shiraki A, Shiraki N, Sakimoto S, Maruyama K, Maeno T, Nishida K. Intraoperative challenges and management of fibrovascular membrane with tractional retinoschisis in proliferative diabetic retinopathy. BMC Ophthalmol 2024;24:1:299.
2. Alshaikhsalama AM, Thompson KN, Patrick H, Lee J, Voor TA, Wang AL. Clinical characteristics and surgical outcomes of patients undergoing pars plana vitrectomy for PDR. Ophthalmol Retina 2024;8:8:823-831.
3. Oshima Y, Wakabayashi T, Sato T, Ohji M, and Tano Y. A 27-gauge instrument system for transconjunctival sutureless microincision vitrectomy surgery. Ophthalmology 2010;117:1: 93–102.
4. Awan MA, Shaheen F, Mohsin F. The anatomical and functional outcomes of 27-gauge pars plana vitrectomy in diabetic tractional retinal detachments in the South Asian population. Cureus 2023;15:4:e38099.
5. Vaz-Pereira S, Dansingani KK, Chen KC, Cooney MJ, Klancnik JM Jr, Engelbert M. Tomographic relationships between retinal neovascularization and the posterior vitreous in proliferative diabetic retinopathy. Retina 2017;37:7:1287-1296.