Take-home points
|
 |
Bio Haley D’Souza, MD, is a medical retina fellow at the Cleveland Clinic Cole Eye Institute in Cleveland, Ohio. |
Over the past three decades, the management of diabetic retinopathy and diabetic macular edema has undergone significant transformation. The introduction of intravitreal anti-vascular endothelial growth factor therapy dramatically improved visual outcomes and redefined standards of care, reducing rates of vision loss and delaying disease progression for millions of patients worldwide.¹
Despite these advances, serial intravitreal anti‑VEGF injections supplemented by focal/grid laser, panretinal photocoagulation or corticosteroids remains the cornerstone of therapy for most patients. In real‑world practice, visit fatigue, treatment cost, systemic comorbidities and logistical barriers make this treatment model particularly challenging for a working‑age population managing chronic diabetes.¹,² Even among adherent patients, a substantial subset of patients demonstrate persistent retinal fluid or limited functional improvement despite adequate VEGF suppression.
These unmet needs have catalyzed a wave of innovation focused not only on improving efficacy, but also on reducing treatment burden and targeting disease mechanisms beyond VEGF. As the field enters the next decade, the DR/DME pipeline increasingly converges on three overarching goals: enhanced durability; expanded mechanisms of action; and more sustainable, accessible care.
Extended‑Delivery and Durable Therapies
The cumulative burden of monthly or bimonthly intravitreal injections affects both patients and retina practices.
On a system-wide level, annual Medicare expenditures for anti‑VEGF therapy have risen substantially, increasing from an estimated $2.51 billion in 2014 to $4.02 billion in 2019.³ Additionally, each injection carries a small but non‑negligible risk of complications such as endophthalmitis, retinal tear or vitreous hemorrhage.4 Therapies that meaningfully extend treatment intervals therefore have the potential to improve access, adherence, and long‑term outcomes, particularly for underserved populations.
The ranibizumab port delivery system (PDS; Susvimo, Genentech/Roche) is currently one of the most mature durability‑focused platforms. It’s a surgically implanted reservoir that enables continuous intravitreal delivery of ranibizumab, with refill exchanges approximately twice yearly.
In May 2025, the PDS received FDA approval for the treatment of both diabetic retinopathy and DME.5 Clinical trial data demonstrate that the PDS can substantially reduce injection frequency while maintaining visual and anatomic outcomes comparable to monthly intravitreal ranibizumab.6,7
Corticosteroid‑based sustained‑release approaches also continue to evolve. In the Phase II RIPPLE‑1 trial, 92 percent of patients treated with the IBE‑814 high‑dose (70 µg) intravitreal dexamethasone insert reached six months without requiring additional therapy.8
Migaldendranib (MGB; Ashvattha Therapeutics) is a VEGF receptor tyrosine kinase inhibitor (TKI) designed to suppress VEGF production from macrophages, microglia, and retinal pigment epithelial cells. Unlike other currently available therapies, it’s administered subcutaneously.
In a recently completed 40‑week Phase II study of MGB in DME and neovascular age‑related macular degeneration, migaldendranib was associated with improvements in both visual acuity and retinal anatomy, along with a marked reduction in supplemental anti‑VEGF injections from an annualized rate of 8.4 injections to 1.6 injections. It was well tolerated, with no reported ocular or systemic serious adverse events despite subcutaneous route of delivery.9
EYP-1901 (Duravyu) is a sustained-release TKI. It’s a bioerodible intravitreal insert that delivers vorolanib, a small-molecule pan-VEGFR inhibitor that also suppresses IL-6/JAK1–mediated inflammatory signaling, achieving rapid therapeutic intraocular levels and maintaining target inhibition for at least six months. In the Phase II VERONA trial in active DME, a single EYP-1901 dose significantly extended time to first supplemental anti-VEGF treatment compared with aflibercept, with 73 percent of eyes treated with the 2.7-mg dose remaining supplement-free through 24 weeks. Early and sustained improvements in visual acuity and retinal thickness were observed, along with reductions in macular vascular leakage and a favorable safety profile.
Pivotal Phase III trials are now underway evaluating q6-month EYP-1901 dosing versus on-label aflibercept, aiming to maintain visual outcomes while meaningfully reducing injection frequency in diabetic macular edema.10
Axpaxli (formerly OTX‑TKI) (axitinib; Ocular Therapeutix), delivered via a biodegradable intravitreal implant, demonstrated encouraging results in moderately severe non-proliferative DR, with no treated eyes progressing to proliferative disease or developing DME at 48 weeks compared with 37 percent of control eyes.11
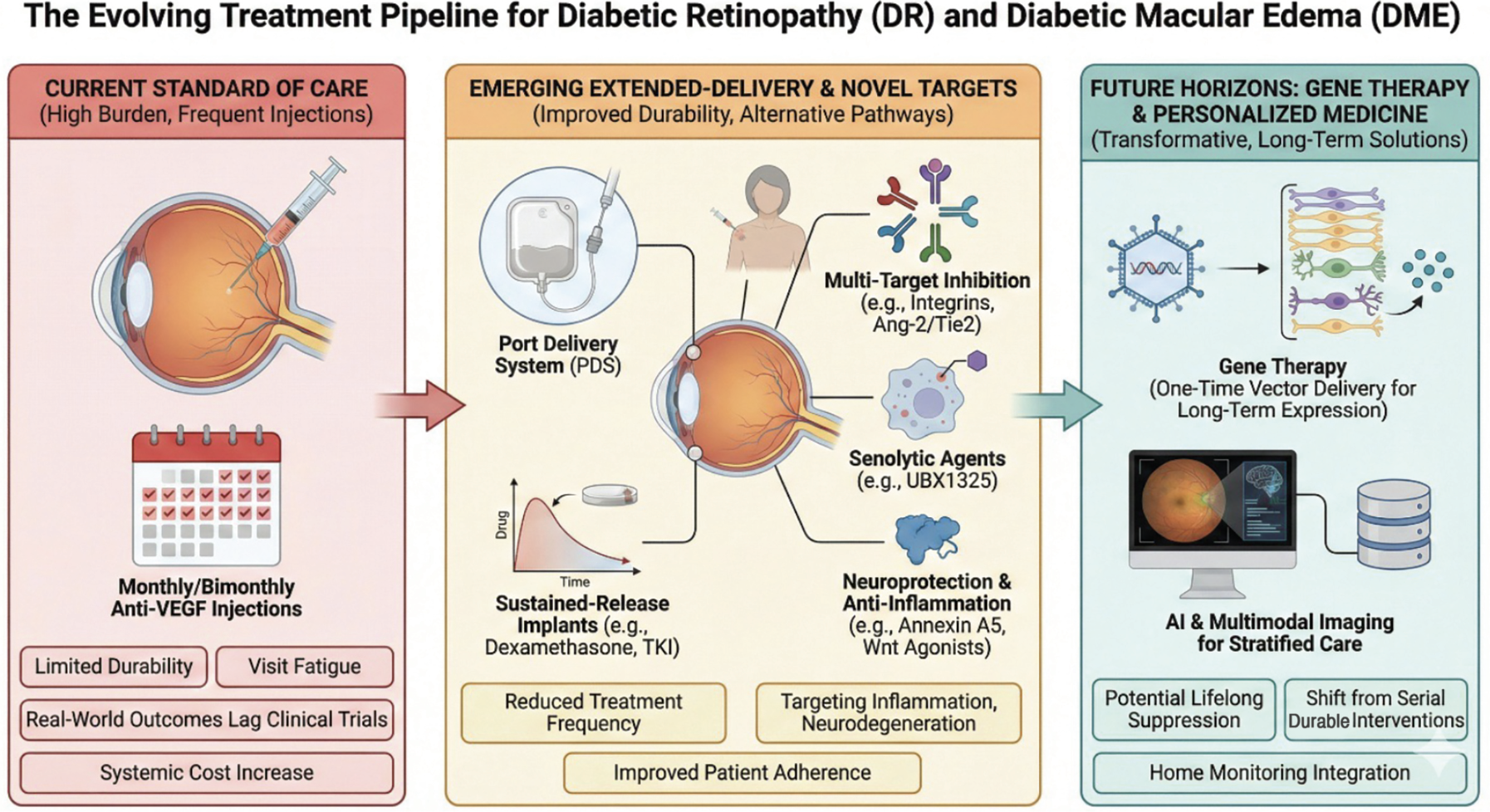 |
| Figure 1. The evolving therapeutic landscape for diabetic retinopathy and diabetic macular edema. The diagram illustrates the progression from the current high-burden anti-VEGF standard of care (left panel) toward emerging extended-delivery systems and novel biologic targets (middle panel), and finally to future gene therapies and AI-driven personalized medicine (right panel). |
Novel Biologic Targets
Diabetic retinal disease is driven by a complex interplay of angiogenesis, inflammation, oxidative stress, vascular dysfunction and neurodegeneration.1 Recognition of this multifactorial pathophysiology has expanded interest in therapeutic targets beyond VEGF inhibition.
OCU200 (Ocugen) is an integrin‑targeting fusion protein that combines tumstatin and transferrin domains to inhibit angiogenesis and vascular leakage through non‑VEGF mechanisms. Preclinical and early clinical studies suggest potential benefit in modulating vascular permeability and inflammation, positioning integrin inhibition as a potentially complementary strategy in diabetic eye disease.12
Additional targeting includes small‑molecule modulation of the Ang‑2/Tie2 pathway and melanocortin receptor agonists, both of which are under investigation for anti‑inflammatory and neuroprotective effects. The Genentech THAMES study (NCT06850922) is a Phase I/II clinical trial evaluating the novel treatment RO7446603 in patients with DME. The study investigates RO7446603 alone or in combination with faricimab, targeting the Tie2 pathway.
ANXV (Annexin Pharmaceuticals), a recombinant annexin A5 protein, is currently being evaluated in a Phase IIa trial for NPDR in patients with early microvascular changes who don’t yet require anti‑VEGF therapy. Annexin A5 is thought to protect endothelial cells, reduce leukocyte adhesion and modulate immune activity. The agent previously demonstrated a favorable safety profile in Phase IIa studies for retinal vein occlusion.13
Cellular senescence has also emerged as a potential contributor to chronic retinal vascular disease. The senolytic agent UBX1325 (foselutoclax; Unity Biotechnology) was evaluated in patients with DME who had shown suboptimal response to anti‑VEGF therapy. Although the study wasn’t powered for efficacy, UBX1325 demonstrated an acceptable safety profile and a numerically greater mean gain in best‑corrected visual acuity, with a 5.6-letter greater mean gain in ETDRS best-corrected visual acuity at week 48 compared to sham.14
Gene Therapy and Transformative Platforms
 |
If extended‑release implants represent an incremental advance, gene therapy offers the possibility of a more transformative shift. Advances in vector engineering and ocular delivery have made long‑term intraocular expression of therapeutic proteins increasingly feasible after a single administration.15
4D‑150 (4D Molecular Therapeutics) employs a proprietary adeno‑associated viral (AAV) vector designed to express dual inhibitors targeting VEGF-A, VEGF-B and placental growth factor. In the Phase III SPECTRA trial in DME, the selected dose (3E10 vg/eye) achieved a 78-percent reduction in treatment burden compared with projected on‑label aflibercept dosing through 60 weeks, with a favorable safety profile and no reported cases of intraocular inflammation, vasculitis or retinal artery occlusion.16,17
Restoret (EYE103; EyeBio/Eyebiotech) is a trispecific Wnt agonist antibody designed to mimic the activity of Norrin, a ligand critical for maintaining the blood–retinal barrier. Preclinical work has demonstrated Norrin‑mediated restoration of barrier integrity and reduction of vascular permeability.18,19
In the Phase Ib/II AMARONE trial, intravitreal Restoret was well tolerated, with no drug‑related serious adverse events reported at 12-week follow‑up. Preliminary data also showed an increase in BCVA of 11.2 EDTRS letters visual acuity and retinal thickness in patients with DME who were concurrently treated with aflibercept.20
ABBV-RGX-314 (RegenxBio, AbbVie) is a subretinal gene therapy consisting of an AAV8 vector containing a gene encoding for a monoclonal antibody fragment designed to neutralize VEGF.
Data from the ATMOSPHERE and ASCENT trials assessing the subretinal delivery of RGX-314 in wet age-related macular degeneration are anticipated in 2026. A Phase III study on the use of suprachoroidal delivery with the in-office SCS Microinjector of ABBV-RGX-314 in DR is being planned.
A one-time gene therapy alternative to frequent intravitreal injections has potential for effectively treating disease, and Phase I/II and Phase II data have demonstrated durability and substantial reduction in treatment burden.21
FT‑003 (Frontera Therapeutics) recently received investigational clearance in China for DR, following earlier development in nAMD and DME. It has shown in trials significant improvements in visual acuity and retinal anatomy, with a marked reduction (or elimination) of the need for anti-VEGF rescue injections over a two-year follow-up.
Clinical studies suggest that a single intravitreal administration may establish sustained intraocular anti‑VEGF expression, with prolonged visual and anatomic benefits and reduced need for rescue injections over extended follow‑up.22
New delivery routes, including suprachoroidal administration, may further refine the safety and efficacy profile of both gene and small‑molecule therapies by limiting anterior‑segment exposure while maintaining chorioretinal bioavailability.
These innovations will require new procedural skill sets, long‑term safety monitoring and thoughtful discussion regarding durability, reversibility and cost.
Looking Ahead
The late‑stage DR/DME pipeline is dynamic. Platforms including port delivery systems, gene therapies and novel targeting pathways exemplify a broader shift toward durable, mechanism‑targeted therapy. Concurrently, artificial intelligence–based image analysis and large‑language‑model tools may enhance disease monitoring and individualized medicine as treatment paradigms grow more complex.
Over the next decade, management of diabetic retinal disease is likely to evolve toward increasingly stratified algorithms. Long‑acting therapies may be deployed earlier to prevent progression, combination or pathway‑specific treatments may be reserved for refractory disease, and gene‑based interventions may ultimately offer multi‑year stability from a single procedure.
Together, these advances promise to reframe DR and DME from conditions managed through relentless retreatment to diseases addressed with durable, precision‑guided interventions. RS
REFERENCES
1. Gonzalez Cortes JH, Fernandez Romero D, Luna Zarate V, et al. Current treatments and innovations in diabetic retinopathy and diabetic macular edema. Front Med (Lausanne) 2022;9:1011658.
2. Lowater SJ, Bhatnagar P, Jain A, et al. Clinical trials and future outlooks of the port delivery system for sustained anti VEGF therapy. Clin Ophthalmol 2023;17:3893 3903.
3. Desai S, Sekimitsu S, Rossin EJ, Zebardast N. Trends in anti vascular endothelial growth factor Original Medicare Part B claims in the United States, 2014 2019. Ophthalmic Epidemiol 2024;31:5:468 477.
4. Ramos MS, Xu LT, Singuri S, et al. Patient-reported complications after intravitreal injection and their predictive factors. Ophthalmol Retina 2021;5:7:625-632.
5. FDA approves Roche’s Susvimo for diabetic retinopathy. Roche Media Release. May 21, 2025. Accessed October 13, 2025.
6. Holekamp NM, Campochiaro PA, Chang MA, et al; Archway Investigators. Archway randomized phase 3 trial of the port delivery system with ranibizumab for neovascular age related macular degeneration. Ophthalmology 2022;129:3:295 307.
7. Khanani AM, Campochiaro PA, Graff JM, et al. Continuous ranibizumab via port delivery system vs monthly ranibizumab for diabetic macular edema: The PAGODA randomized clinical trial. JAMA Ophthalmol 2025;143:4:326 335.
8. Wootton K, Mackey G, Simpson M, et al. IBE 814 intravitreal implants reduce treatment burden in subjects with DME and RVO: RIPPLE 1 phase 2 trial. Invest Ophthalmol Vis Sci 2024;65:3121.
9. Hennings D. Ashvattha Therapeutics announces positive topline 40 week phase 2 results for migaldendranib. Ashvattha Therapeutics. September 4, 2025. Accessed October 13, 2025.
10. Singer M, VERONA Investigators. VERONA: Results from a Phase 2 Trial of EYP-1901 (Vorolanib Intravitreal Insert) versus Aflibercept for Diabetic Macular Edema. https://eyepoint.bio/wpcontent/uploads/2025/07/MaculArt VERONA Singer Draft 25June2025 FINAL.pdf. Accessed February 6, 2026.
11. Ocular Therapeutix news. Ocular Therapeutix announces positive topline Phase 1 data for AXPAXLI in Diabetic Retinopathy. https://investors.ocutx.com/node/12691/pdf. Accessed February 6, 2026.
12. Ocugen news release. https://ir.ocugen.com/news-releases/news-release-details/ocugen-inc-announces-first-patient-dosed-phase-1-clinical-trial/. Accessed February 6, 2026.
13. Annexin Pharmaceuticals. Annexin receives approval for phase 2a study in diabetic retinopathy and RVO. August 27, 2025. Accessed October 13, 2025.
14. Klier S, Dananberg J, Masaki L, et al. Safety and efficacy of senolytic UBX1325 in diabetic macular edema. NEJM Evid 2025;4:5:EVIDoa2400009.
15. Rowe LW, Ciulla TA. Gene therapy for non hereditary retinal disease. Genes (Basel) 2024;15:6:720.
16. 4D Molecular Therapeutics. 4DMT presents positive 60 week results from 4D 150 SPECTRA clinical trial. July 31, 2025. Accessed October 13, 2025.
17. ClinicalTrials.gov. 4D 150 in patients with diabetic macular edema. Updated September 22, 2025. Accessed October 13, 2025.
18. Díaz Coránguez M, Lin CM, Liebner S, Antonetti DA. Norrin restores blood retinal barrier properties. J Biol Chem 2020;295:14:4647 4660.
19. Chidiac R. A Norrin/Wnt surrogate antibody stimulates endothelial cell barrier function and rescues retinopathy EMBO Mol Med 2021;13:7:e13977.
20. BioSpace. EyeBio announces first patients dosed in phase 1b/2 trial of Restoret. 2023.
21. AbbVie. AbbVie and REGENXBIO announce updates on the ABBV RGX 314 clinical program. January 13, 2025. Accessed October 13, 2025.
22. Frontera Therapeutics. Frontera Therapeutics Announces IND Clearance for FT-003 in Diabetic Retinopathy. https://fronteratherapeutics.com/news/223.html. March 13, 2025. Accessed December 8, 2025.