Take-home points
|
 |
Bio Dr. Bowe is a second year vitreoretinal surgery fellow at Mid-Atlantic Retina/Wills Eye Hospital |
Recurrent rhegmatogenous retinal detachment in the absence of proliferative vitreoretinopathy is typically a mechanical process. This is related to new or missed breaks, inadequate treatment of the pathology, persistent vitreoretinal traction or inadequate tamponade. Notably, re-detachments due to the cellular proliferation and traction of PVR are a distinct topic.
The risk of re-detachments has decreased with advances in vitrectomy techniques; however, it still represents a significant issue for vitreoretinal surgeons and patients. Recent studies have noted that recurrent RRD occurs in approximately 2.5 to 8 percent of cases within the United States and 16 to 28 percent globally. The presence of PVR increases the recurrence rate to 25 to 33 percent, with increased rates associated with grade C PVR or worse.1-5
This review summarizes the causes, prevention, and management of recurrent retinal detachments in the absence of PVR.
Etiology
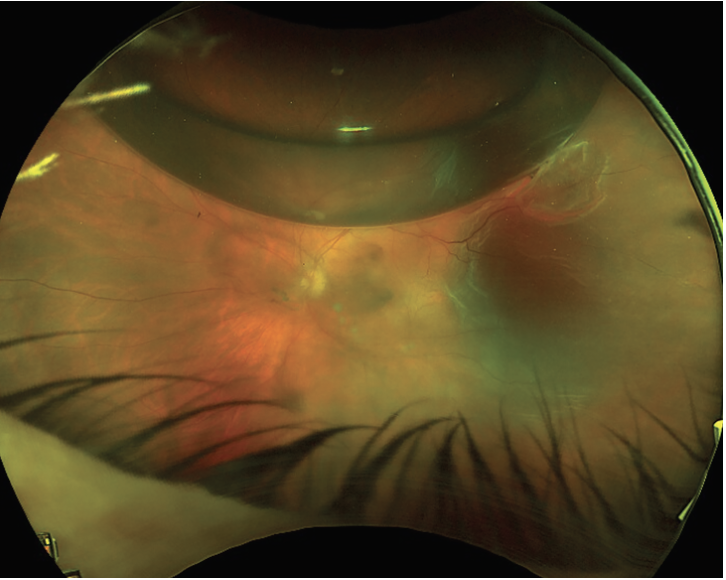 |
| Figure 1. Recurrent retinal detachment caused by a new tear (2:00) in an eye with a previously well treated retinal detachment (with a break at 11:00). |
Recurrent RRD without PVR is a mechanical process. Differentiation between the two types of re-detachments is critical. PVR can be characterized as a maladaptive wound healing process associated with cellular proliferation, membrane formation, preretinal, intraretinal and subretinal fibrosis and contraction, which can lead to recurrent RRD. Re-detachments in the setting of PVR require a different approach (possibly adding a scleral buckle, as well as early intervention in PVR RRDs).
The causes of recurrent RRD in the absence of PVR can be separated into the following categories, and there may be more than one cause in a single case:
• Missed retinal breaks. Small tears or retinal defects may be overlooked during the initial surgery, particularly in eyes where the view is compromised by media opacity.
• Persistent vitreoretinal traction. Residual vitreoretinal traction on the causative break may allow continual accumulation of subretinal fluid.
• New break development. Continued vitreous traction or vitreous contraction can lead to the development of new breaks (Figure 1).
• Inadequate retinopexy. Inadequate sealing of the breaks (pexy, or positioning/height of a buckle) may lead to recurrent accumulation of subretinal fluid.
• Inadequate tamponade. Inadequate tamponade (short-acting, insufficient fill, etc) may fail to support the break until sufficient chorioretinal adhesion is achieved.
Risk factors for re-detachment include:
• Surgical and anatomic factors. Missed breaks (often in the setting of lattice and/or thin peripheral retina), inadequate vitrectomy, inadequate scleral depression, inadequate pexy or tamponade, or poor selection of method of surgical repair.
• Patient factors. Inadequate positioning due to various reasons.
Evaluation
Differentiation between PVR RRD and recurrent RRD without PVR is critical, as mentioned above, due to the differing approaches to fixing them.
A thorough history and examination (which may include fundus photography and OCT, and should include a detailed scleral-depressed examination) targeted at understanding the initial method of repair and the reason for the recurrent RRD must be undertaken.
Management
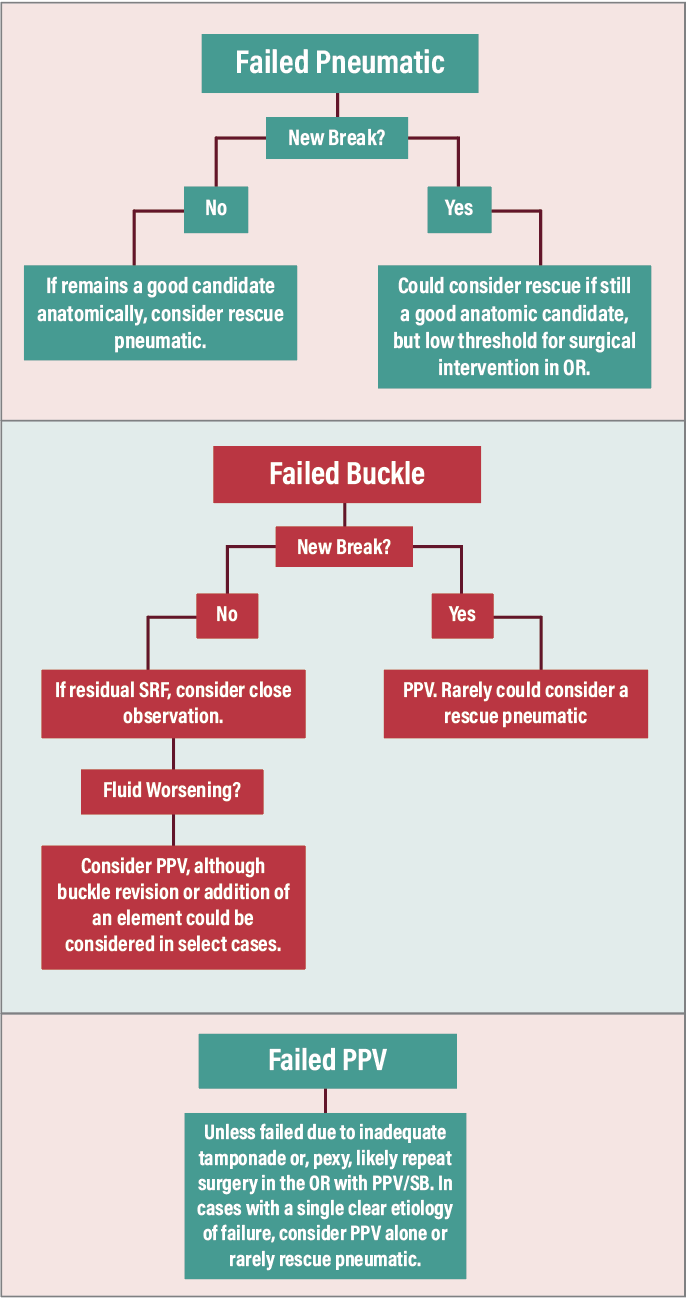 |
| A flowchart for approaching recurrent retinal detachments. Decisions are usually made on a case-by-case basis, however. |
As with any complication or unanticipated outcome, prevention is key. To increase the success rate, a surgeon should be competent in all methods of detachment repair. Although there has been a significant trend towards pars plana vitrectomy in the treatment of retinal detachment,6 scleral buckling and pneumatic retinopexy remain crucial techniques that have their own advantages. Vitreoretinal Surgery Fellowships should, therefore, ensure trainees are skilled in all these approaches.
Despite our best efforts, recurrent detachments without PVR still occur. The first step to managing a recurrent RRD is correctly identifying the cause (detailed above), as this will guide appropriate treatment.
In cases of failed primary PPV, we have a low threshold to add an encircling scleral buckle during repeat PPV. SB improves success rates in phakic patients and potentially in other cases, such as those with inferior retinal detachments.7 Although there is conflicting data regarding improved success rates in non-phakic eyes,7,8 scleral buckling treats persistent peripheral vitreoretinal traction. Additionally, it can support other pathology,such as small peripheral holes or lattice degeneration, which may have been missed in the primary surgery. Adding a buckle is rarely regretted, especially in cases that have already failed one vitrectomy surgery. Meticulous scleral depression is also important to identify and treat all potential pathology. Longer tamponades such as C3F8 or silicone oil may also be more beneficial, particularly in cases with inferior pathology, which may not be adequately supported with SF6. This is determined on a case-by-case basis.
Similarly, when a primary buckle fails to treat a retinal detachment, the cause must be determined so that it can be adequately addressed. Of note, subretinal fluid can take weeks to months to resorb, and this is not considered a failure. We observe these cases, particularly when there is continued resolution of subretinal fluid and the pathology is supported by the buckle. However, if the initial break isn’t supported, new breaks occur, or subretinal fluid is worsening, further intervention is necessary. The procedure will depend on both the patient’s exam and the surgeon’s skill set. Occasionally, the buckle can be revised if the surgeon feels comfortable doing so. This is particularly useful in younger patients or in those without a posterior vitreous detachment, in which case a vitrectomy may be more challenging.
You can add SB elements if additional areas need to be supported, or the buckle (such as an encircling band) can be repositioned if the original pathology isn’t well supported. For other cases, or if the surgeon feels more comfortable doing so, they can perform a PPV. It’s important to identify and treat all pathology as previously detailed above. During a vitrectomy, it may be reasonable to apply peripheral laser along the extent of the detachment if suspected pathology is present. Notably, even a rescue pneumatic retinopexy could also be considered in select cases where the characteristics of the detachment and the patient selection are both favorable.
The same algorithm can apply if the initial procedure was a PnR. Patient selection is paramount in considering PnR. An excellent candidate would be phakic, with a break confined to the superior eight clock-hours of retina, without PVR and with a good ability to adhere to positioning. Alternatively, if there are multiple breaks, they should be confined within one clock-hour. Also, any lattice degeneration, or retinal holes/tears should be pretreated with laser or cryopexy.9 These can still fail because of inadequate pexy, positioning or tamponade. A rescue pneumatic occasionally can be a reasonable choice, if inadequate positioning wasn’t the cause of failure. At times, displacement of gas into the anterior chamber is responsible for failure. These can be minimized by optimized head positioning during paracentesis and by minimizing pupil diameter. If rescue pneumatic fails or is inappropriate, escalation to either PPV, SB or PPV/SB should be done promptly. The residual gas bubble can be used to keep the macula attached with facedown positioning while awaiting operative intervention.
Effective management of recurrent RRD requires first determining the underlying cause and understanding how the initial repair may have contributed to failure. Treatment decisions are tailored to the location and number of breaks, adequacy of prior tamponade and positioning, and the presence of factors such as lattice degeneration or high myopia. Depending on these variables, numerous interventions may be suggested. Regardless of the chosen method, success hinges on identifying all breaks, ensuring thorough vitreous removal when performing vitrectomy and applying durable retinopexy to achieve long-term reattachment. Each case must be analyzed individually to come to the best management plan.
Outcomes and prognosis
 |
Recurrent RRD without PVR generally carries a more favorable prognosis than PVR-associated failures, provided that the macula was attached during most of the recurrence, the duration of detachment was short and the causative break or tractional source is accurately identified.
Visual recovery is strongly influenced by macular status; eyes with macula-on recurrence or brief macular detachment often return close to baseline. Importantly, the risk of subsequent recurrence after appropriate re-intervention is low, particularly when a buckle is added in anatomically high-risk cases.
Conclusion
Recurrent RRD in the absence of PVR remains primarily a mechanical problem, and successful management requires careful differentiation from PVR-associated failure. A methodical approach focusing on identifying all breaks, eliminating traction, applying durable retinopexy and providing adequate tamponade can yield excellent anatomic outcomes. With modern surgical techniques and proper case selection, most patients can achieve retinal reattachment with meaningful visual recovery. RS
REFERENCES
1. Momenaei B, Wakabayashi T, Kazan AS, et al. Incidence and outcomes of recurrent retinal detachment after cataract surgery in eyes with prior retinal detachment repair. Ophthalmology Retina 2024;8:5:447-455.
2. Stenz EC, Yu HJ, Shah AR, et al. Outcomes of eyes undergoing multiple surgical interventions after failure of primary rhegmatogenous retinal detachment repair. Ophthalmology Retina. 2022;6:5:339-346.
3. Tawfik AM, Eweidah AM, Hassanien RA, Mohamed SF, Kasem RA, Ghoneem M. Incidence and risk factors for recurrence after surgical treatment of rhegmatogenous retinal detachment: A retrospective cohort study. Int J Retina Vitreous 2025;11:1:59.
4. Irigoyen C, Goikoetxea-Zubeldia A, Sanchez-Molina J, Amenabar Alonso A, Ruiz-Miguel M, Iglesias-Gaspar MT. Incidence and risk factors affecting the recurrence of primary retinal detachment in a tertiary hospital in Spain. Journal of Clinical Medicine 2022; 11:15:4551.
5. Peck TJ, Starr MR, Yonekawa Y, et al. Outcomes of primary rhegmatogenous retinal detachment repair in eyes with preoperative grade B or C proliferative vitreoretinopathy. J Vitreoretin Dis 2021;6:3:194-200.
6. Reeves, M-G, Pershing, S, Afshar, AR. Choice of primary rhegmatogenous retinal detachment repair method in US commercially insured and Medicare Advantage patients, 2003-2016. American Journal of Ophthalmology 2018;196:82–90.
7. Starr MR, Ryan EDH, Yonekawa Y. Primary retinal detachment outcomes study: Summary of reports number 1 to number 18. Curr Opin Ophthalmol 2023;34:211-217
8. Hebert M, Garneau J, Doukkali S et al. Retina 2024, 44 :1899-1904.
9. Juncal VR, Bamakrid M, Jin S, et al. Pneumatic retinopexy in patients with primary rhegmatogenous retinal detachment meeting PIVOT trial criteria. Ophthalmol Retina 2021;5:3:262-269.