Take-home points
|
 |
Bios Dr. Muste is a vitreoretinal surgical fellow at Wills Eye Hospital in Philadelphia. Dr. Garg is a professor of Ophthalmology on the Retina Service at Wills Eye Hospital and Thomas Jefferson University, and a partner at Mid Atlantic Retina. He also serves as co-director of the Retina Research Unit at Wills. Dr. Hsu is a faculty member of the Retina Service at Wills Eye Hospital and professor of ophthalmology at the Sidney Kimmel Medical College of Thomas Jefferson University. He serves as co-director of the Retina Research Unit at Wills. |
Despite advances in vitreoretinal surgical techniques, secondary epiretinal membrane formation occurs after retinal tears as well as after rhegmatogenous retinal detachment. After retinal tears, ERM occurs in 2.9 to 31.7 percent of patients. After RRD, the incidence ranges from 4.6 percent to as high as 49 to 70.3 percent depending on detection methods, patient characteristics and follow-up duration.1-4 Thankfully, not all ERMs are visually significant.4
A clinically significant subset of epiretinal membranes develop rapidly in the early postoperative period, leading to metamorphopsia, decreased visual acuity and patient dissatisfaction despite successful retinal detachment repair.
The challenge for retinal surgeons lies in identifying which patients will develop problematic membranes and determining the optimal timing for surgical intervention in these cases.
When do hyperacute ERMs develop?
The term “hyperacute” distinguishes rapidly forming postoperative ERMs from more indolent idiopathic membranes. A hyperacute membrane develops within the first three months following repair, with approximately 90 percent of ERMs following RD appearing during this window.1,5
Ishida and colleagues performed systematic optical coherence tomography surveillance of 322 eyes at one, three, six and 12 months after retinal detachment repair. Of the 39 eyes that developed an ERM over the 12-month follow-up period, 13 eyes (33.3 percent) developed an ERM by one month, 30 eyes had an ERM at 3 months (77 percent) and 36 eyes had an ERM at six months (92.3 percent). Notably the study was anatomic in scope and visual significance wasn’t discussed; most likely many of these ERMs weren’t visually significant.
There’s less literature addressing ERM that occurs after laser retinopexy for horseshoe tear. Blackorby and colleagues showed 2.9 percent of eyes developed an ERM after LR. The average time to ERM development was 12 months after laser retinopexy (Range: 1.5 to 94 months).6 In a series of clinical outcomes of HST after requiring LR, Choi and colleagues incidentally noted 7.4 percent of eyes developed an ERM at three months.7 However, no single study specifically addresses hyperacute ERM following LR. As noted above, it’s not clear how visually significant the ERMs are.
From our experience, visually significant ERM can form after any retinal break including HST alone (Figure 1) and RD (Figure 2) within 90 days. However, future studies are needed to characterize the timing and risk factors for membrane formation.
Why membranes form: Pathophysiology and risk factors
ERM derives from liberated retinal pigment epithelial cells undergoing transformation.5,8 Factors such as larger or more numerous tears and more extensive RDs are associated with increased RPE cell liberation.
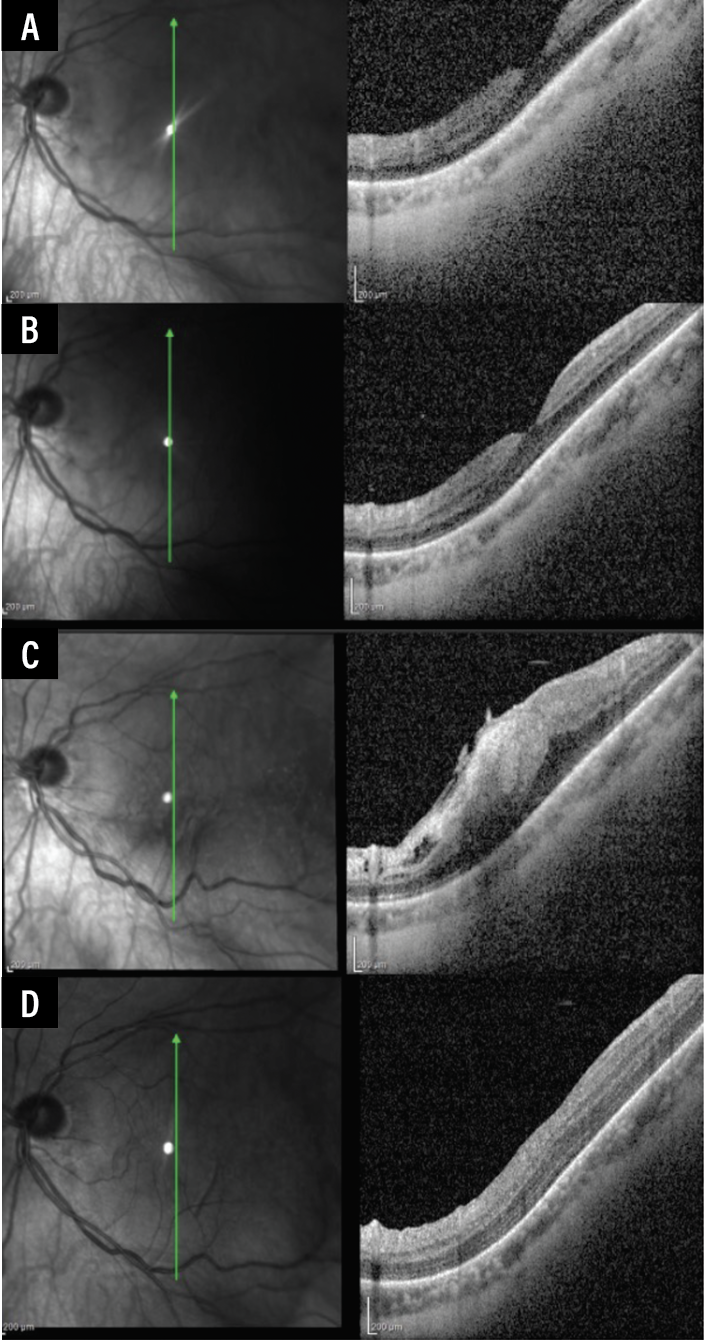 |
| Figure 1. (A) Vertical raster of OCT macula of a patient presenting for a new HST with a visual acuity of 20/30. (B) Two weeks after laser retinopexy, the OCT macula remained unchanged and the visual acuity remained 20/30. (C) Five weeks after laser retinopexy, visual acuity declined to 20/100 with ERM formation. (D) The patient was taken for ILM peel with OCT two months after presentation. One month after surgery, there was restoration of foveal architecture. Although the patient reported improved vision quality, the vision recovery wasn’t complete at this point, with vision at 20/350 and improving. |
Hirakata and colleagues identified four perioperative risk factors that increase the odds of ERM formation: postoperative redetachment (OR 19.7; 95% CI [4.87–79.38], p<0.01), retinal detachment area >3 clock hours (OR 12.91; 95% CI [2.34–71.19] p< 0.01), more than three retinal breaks (OR 8.07; 95% CI [2.35–27.71],p<0.01) and preoperative vitreous hemorrhage (OR 4.71; 95% CI [1.19–18.62], p=0.03).9 Other authors similarly underscore the importance of these risk factors, which may elevate inflammatory mediators and increase cellular proliferation.1,5,8,9
Intraoperative factors matter as well. For ERM in general, Szigiato and colleagues report laser photocoagulation exceeding 1,000 spots, choroidal detachment and 360-degree laser treatment all significantly increase ERM formation risk after PPV for RD repair.8 The data around cryotherapy is mixed with some authors finding a significant association with ERM formation while others report no association.8,9 Some authors suggest that initial macula-off status increases the likelihood of developing an ERM while others haven’t found any association.1,2,4,8
Israelivich and colleagues examined 42 eyes that underwent ERM surgery after LR. They found 16 of 42 eyes (38.1 percent) that underwent surgery ≤180 days from LR were younger and more likely to have Stage 4 ERM characteristics with ectopic inner foveal layers and disorganized retinal layers.10 Notably, the presence of VH at the time of laser, number of retinal tears and location of retinal tear weren’t different in eyes the ≤180 days versus >180 day group. However, concurrent RD with a mean of 1.7 clock hours in size was more likely to result in earlier surgery for ERM. This is in line with the above discussion regarding liberated RPE as a driver of ERM formation.
Seeing Cellophane: OCT classification and staging
Spectral-domain OCT has fundamentally changed how we detect and characterize ERMs. Although the classification system developed by Govetto and colleagues was originally used for idiopathic membranes, it provides a standardized framework that can be used in hyperacute membranes as well.11
The classification progresses through four stages:
— Stage 1 membranes preserve the foveal depression with well-defined retinal layers.
— Stage 2 demonstrates loss of the foveal pit while maintaining distinguishable retinal architecture.
— Stage 3 ERMs have EIFL in addition to Stage 2 features.
— Stage 4 ERM show EIFL with disrupted and disorganized retinal layers.11
Some authors report 65 to 67 percent of ERMs that occur within three months after RRD repair and LR reach Stage 4.1,12 It’s known that EIFL and disruption of outer retinal structures correlates with worse visual outcomes and persistent metamorphopsia. These findings raise the question of whether intervention before structural changes become irreversible is better than waiting for more vision loss to occur. However, these studies often select for eyes undergoing surgery for ERM repair. As such it’s difficult to be certain what proportion of ERMs that present following RRD or LR will advance to Stage 4 when considering the whole population.
When to intervene?
Ultimately, this question is best resolved on a case-by-case basis. A visually significant cataract may need to be removed before or concurrent with PPV and membrane peeling for a visually significant ERM. Proceeding with combined surgery or staging is a separate area of discussion that we will save for a future article.
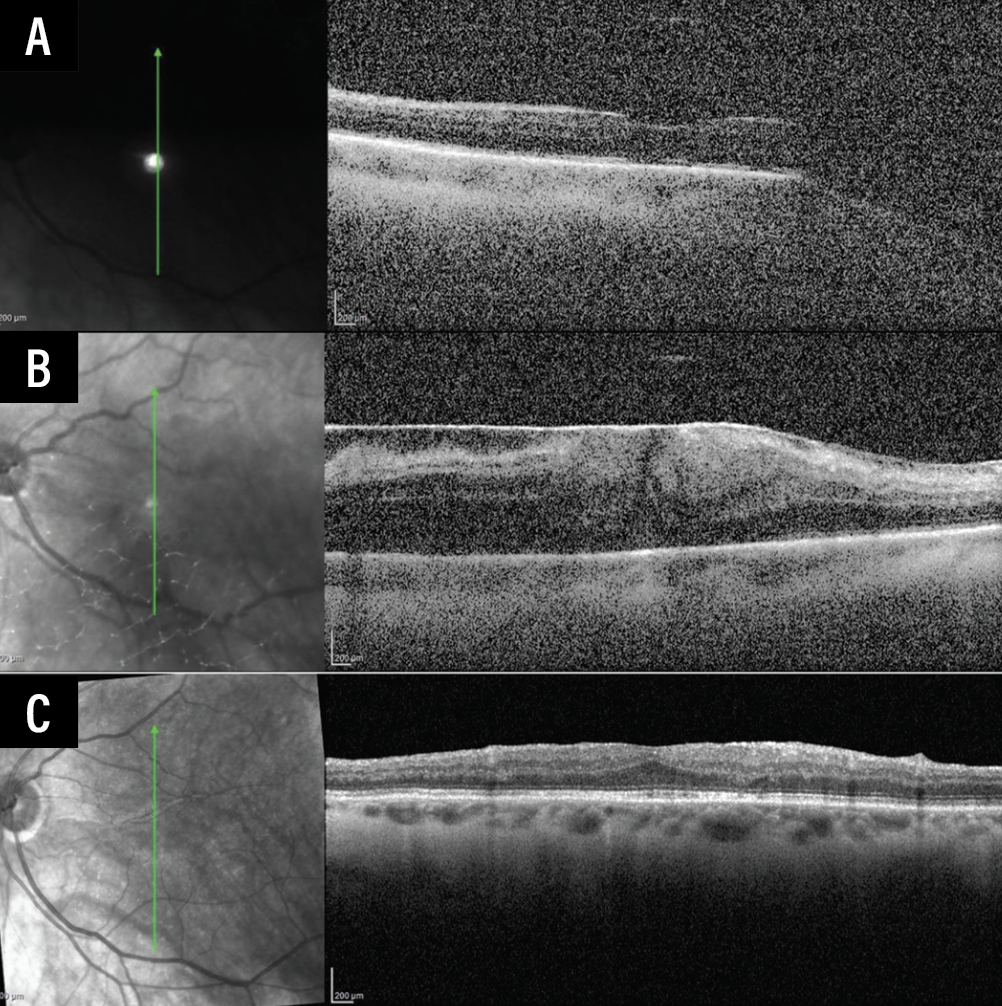 |
| Figure 2. (A) Vertical raster of OCT macula of the left eye one month after pars plana vitrectomy for retinal detachment repair. Visual acuity was 20/150, an improvement from the presenting visual acuity of counting fingers. (B) Eight weeks after retinal detachment repair, an ERM had formed with the visual acuity declining to counting fingers. (C) The patient had an ILM peel 12 weeks after retinal detachment repair. One year following ILM peeling, the OCT continued to improve with reconstitution of foveal architecture with the visual acuity improving to 20/40. |
Regarding hyperacute ERM, there’s some data that can guide management. Bomdica and colleagues report a series of 55 cases of ERM following RRD repair. The authors divided the group into 37 eyes undergoing PPV and ILM peel within six months and 18 eyes treated beyond six months. These groups achieved a final visual acuity of 20/40 and 20/80, respectively.1 The authors suggest patients who had a membrane peel six months or later had cataract surgery prior to the PPV for the ERM and this was likely the cause for the delay. The authors suggest earlier intervention (within six months following RD occurrence as per their study design) may be beneficial. This fits with the pathophysiology and staging reviewed above. Presumably, the more time that an aggressive, rapidly progressive ERM can exert traction on the macula, the more likely macula dysfunction occurs.
We favor earlier intervention in these cases as anecdotally we seem to achieve better visual and anatomic outcomes (Figures 1 and 2).
PPV with ILM peel during the ERM surgery rather than ERM peel alone also seems to be better. Council and colleagues reported positive outcomes with no significant increase in recurrent retinal redetachment following ILM peel. It should be noted that 79.1 percent of the phakic patients in their study underwent subsequent phacoemulsification.13 As such, all patients should be counseled about cataract formation after vitrectomy.
Following cataract surgery, patients may be advised of the risks of macular edema and macular hole formation. Mylonas and colleagues found higher rates of macular edema up to 42 percent following successful cataract surgery in eyes with PPV and ILM peel.14 Though no study clearly tracks the rate of macular hole formation following cataract surgery in eyes with PPV and ILM peel, Rush and colleagues retrospectively reviewed 423 eyes after PPV and ILM peel. They found macular holes may form in 2.6 percent of cases.15
What about peeling at the time of initial RRD repair?
The rationale for ILM peeling prophylactically at the time of RRD is straightforward: If there’s a risk for ERM formation, why not remove the scaffold on which membranes develop? ILM peeling during RD repair reduces postoperative ERM formation. A recent meta-analysis of 3,420 eyes found ILM peeling reduced ERM formation to 1.5 percent from 8.5 percent in the control group. However, no significant difference in mean visual acuity was noted between the two groups, suggesting that waiting for the ERM to occur and fixing it at that point is a reasonable option.16
As discussed above, not all ERMs are visually significant, and vitrectomy with membrane peel can lead to iatrogenic retinal breaks, inner retinal dimpling and retinal pits. These may be signs of Mueller cell trauma and correspond to microscotoma, which raises questions of subclinical effects of peeling.17,18 One potential limitation of this study is that the final follow-up varies among studies and this may affect how authors appreciate final visual improvement. For example, Nam and colleagues observed eyes to 12 months after peeling and reported a better mean visual acuity in ILM peel patients.19 Similarly, cataract management also varied between the studies and could have influenced rates.
Currently, we don’t routinely peel ILM in eyes undergoing RRD repair. However, selective prophylaxis in high-risk cases is worth considering. Patients with preoperative vitreous hemorrhage, extensive laser treatment (>1,000 spots) or preexisting retinal surface wrinkling during surgery may benefit most from prophylactic intervention.
 |
What is the optimal management strategy?
We recommend a risk-stratified approach to managing hyperacute ERM after retinal tears with or without a history of RD repair.
• Routine-risk cases. Implement surveillance with OCT imaging at one month and three months postoperatively to capture the peak window of ERM development. Most patients will not develop visually significant membranes that require intervention, but they should be counseled about doing monocular vision checks regularly.
• High-risk cases. Consider prophylactic ILM peeling during the primary repair. The appearance of retinal surface wrinkling deserves particular attention. Akiyama and colleagues found that specifically peeling eyes that had visible wrinkling prevented all visually significant postoperative ERMs, though this finding may reflect early PVR-B changes.3,18
• Symptomatic ERMs developing during surveillance. Consider intervention within six months of RD/tear repair. Waiting for cataract maturation or adhering to arbitrary time intervals may be unnecessary and may compromise outcomes. Some eyes have profound ERM formation that looks and behaves like extensive PVR. In these instances, it may be prudent to perform MP early.
• Counsel phakic patients. Cataract surgery will likely be needed after vitrectomy.13 In selected cases and certain localities, combined cataract surgery with membrane peeling may be preferred.
• Not everyone with an ERM needs surgery. In Szigiato and colleagues’s cohort, 58 percent of patients developed OCT-visible ERMs but only 5 percent ultimately required surgical intervention.8 This underscores the importance of clinical correlation and patient symptoms rather than treating OCT findings alone.
What patients can expect: Outcomes and prognosis
Setting appropriate expectations is crucial. Visual prognosis following PPV with membrane peeling depends on multiple factors: membrane stage and duration, integrity of outer retinal structures and baseline visual potential.
From the literature regarding idiopathic ERMs, metamorphopsia persists in a substantial proportion of patients despite anatomic improvement, particularly in eyes with preoperative ectopic inner foveal layers.11 While visual acuity typically improves following membrane peeling, some degree of distortion often remains. Patients should understand that symptomatic improvement may take months, and some metamorphopsia and blurriness can persist.
Council and colleagues’s series of 79 eyes undergoing ERM peeling after RD repair showed that 79 percent achieved visual acuity of 20/40 or better, though 21 percent had persistent vision of 20/50 or worse.13 With this data in mind, it might be prudent to emphasize that membrane peeling aims to halt progression and improve symptoms rather than restore normal vision. Moreover, the original RRD may have already caused permanent changes that limit final visual potential regardless of subsequent membrane surgery.
Looking ahead
Although controlling for individual features of RRD and HST isn’t easily done, there are opportunities for future research. Potential areas of study could include identifying OCT biomarkers to predict significant ERMs, refining patient selection criteria for prophylactic ILM peeling and developing adjunctive therapies to modulate the inflammatory cascade driving membrane formation.
Until such advances materialize, the combination of vigilant surveillance, individualized surgical decision-making and timely intervention when indicated remains our best approach to optimizing outcomes for patients developing hyperacute ERMs after RRD repair. RS
REFERENCES
1. Bomdica PR, Shepherd EA, Gupta R, et al. Timing of epiretinal membrane surgery after uncomplicated retinal detachment repair. J Vitreoretin Dis 2025;24741264251337107
2. Sella R, Sternfeld A, Budnik I, et al. Epiretinal membrane following pars plana vitrectomy for rhegmatogenous retinal detachment repair. Int J Ophthalmol 2019;12:12:1872-1877
3. Akiyama K, Fujinami K, Watanabe K, et al. Retinal surface wrinkling as an indicator for internal limiting membrane peeling during vitrectomy for retinal detachment. Retina 2021;41:8:1618-1626
4. Matoba R, Kanzaki Y, Doi S, et al. Assessment of epiretinal membrane formation using en face optical coherence tomography after rhegmatogenous retinal detachment repair. Graefes Arch Clin Exp Ophthalmol 2021;259:9:2503-2512
5. Ishida Y, Iwama Y, Nakashima H, et al. Risk factors, onset, and progression of epiretinal membrane after 25-gauge pars plana vitrectomy for rhegmatogenous retinal detachment. Ophthalmol Retina 2020;4:3:284-288
6. Blackorby BL, Jeroudi AM, Blinder KJ, et al. Epiretinal membrane formation after treatment of retinal breaks: Cryoretinopexy versus laser retinopexy. Ophthalmol Retina 2019;3:12:1087-1090
7. Choi S, Goduni L, Wald KJ. Clinical outcomes of symptomatic horseshoe tears after laser retinopexy. J Vitreoretin Dis 2023;7:4:290-292
8. Szigiato AA, Antaki F, Javidi S, et al. Risk factors for epiretinal membrane formation and peeling following pars plana vitrectomy for primary rhegmatogenous retinal detachment, an OCT guided analysis. Int J Retina Vitr 2022;8:1:70
9. Hirakata T, Hiratsuka Y, Yamamoto S, et al. Risk factors for macular pucker after rhegmatogenous retinal detachment surgery. Sci Rep 2021;11:1:18276
10. Israilevich R, Salabati M, Mahmoudzadeh R, et al. Secondary epiretinal membrane after laser retinopexy for retinal tear or localized retinal detachment: Surgical outcomes and optical coherence tomography structural analysis. Retina 2022;42:1:38-45
11. Govetto A, Lalane RA, Sarraf D, et al. Insights into epiretinal membranes: Presence of ectopic inner foveal layers and a new optical coherence tomography staging scheme. Am J Ophthalmol 2017;175:99-113
12. Shepherd A, Chaturvedi V. Epiretinal membrane surgery within 1 year of retinal detachment repair: Clinical outcomes & anatomic characteristics. Invest Ophthalmol Vis Sci 2023;64:8:5249-5249
13. Council MD, Shah GK, Lee HC, et al. Visual outcomes and complications of epiretinal membrane removal secondary to rhegmatogenous retinal detachment. Ophthalmology 2005;112:7:1218-1221
14. Mylonas G, Sacu S, Deák G, et al. Macular edema following cataract surgery in eyes with previous 23-gauge vitrectomy and peeling of the internal limiting membrane. Am J Ophthalmol 2013;155:2:253-259.e2
15. Rush RB, Simunovic MP, Aragon AV, et al. Postoperative macular hole formation after vitrectomy with internal limiting membrane peeling for the treatment of epiretinal membrane. Retina 2014;34:5:890-896
16. Lamas-Francis D, Bande-Rodríguez M, Blanco-Teijeiro MJ. Primary ILM peeling during retinal detachment repair: A systematic review and meta-analysis. Sci Rep 2023;13:3586
17. Deltour JB, Grimbert P, Masse H, et al. Detrimental effects of active internal limiting membrane peeling during epiretinal membrane surgery: Microperimetric analysis. Retina 2017;37:3:544-552
18. Eckardt F, Klaas J, Siedlecki J, et al. Internal limiting membrane peeling in primary rhegmatogenous retinal detachment: Functional and morphologic results. Klin Monatsbl Augenheilkd 2025;242:2:153-159
19. Nam KY, Kim JY. Effect of internal limiting membrane peeling on the development of epiretinal membrane after pars plana vitrectomy for primary rhegmatogenous retinal detachment. Retina 2015;35:5:880-885.