|
Bio Dr. Thomas is a partner in vitreoretinal surgery and uveitis at Tennessee Retina in Nashville.Disclosures: No financial interests to disclose. |
Managing uveitis in pregnancy requires a thoughtful balance between controlling intraocular inflammation to preserve maternal vision and minimizing potential risks to the developing fetus. Pregnancy is a unique physiologic state characterized by substantial hormonal and immunologic shifts, and these changes have important implications for autoimmune and inflammatory diseases, including uveitis. Treatment decisions must simultaneously consider disease subtype, maternal health, fetal safety, medication pharmacology and the timing of exposure. The result is a clinical landscape defined by nuance, shared decision-making and individualized care.
This article summarizes practical principles for evaluating and managing uveitis during pregnancy, integrating evidence-based guidance with real-world considerations commonly encountered in ophthalmology and maternal-fetal medicine practices.
Physiologic and immunologic changes in pregnancy
Pregnancy induces a shifting immunologic environment that’s broadly immunosuppressive but highly dynamic. As early as the peri-fertilization period, hormonal and immune modulation shape maternal immune tolerance. Although pregnancy is often described as favoring immune suppression, autoimmune and inflammatory diseases behave inconsistently: Some improve, some remain stable and others worsen.1-8
Uveitis often follows a characteristic trend through each stage of pregnancy and immediately after.
- First trimester: This is highly variable on an individual basis. Some patients flare, some improve and some see no change. Early pregnancy may reflect pre-pregnancy disease activity more than pregnancy physiology.
- Second and third trimesters: Many forms of non-infectious uveitis tend to improve as pregnancy progresses and immunologic tolerance increases.
- Postpartum period: Flares are common due to rebound immune activation.
However, not all uveitis subtypes follow the same pattern. Certain groups—particularly HLA-B27-associated acute anterior uveitis and scleritis, especially when related to systemic lupus erythematosus—may worsen during pregnancy. These variants require closer monitoring and proactive management.
Overall, a patient’s degree of disease control prior to conception remains the best predictor of flare risk during pregnancy.
Medication safety: Understanding the framework
Historically, clinicians relied heavily on the FDA’s pregnancy risk categories (A, B, C, D, X), but this system was discontinued in 2015 because of oversimplification. Many effective and commonly used medications, including steroids, insulin and antibiotics, were categorized as C or D simply due to lack of controlled data, not because they were unsafe. Clinicians should now rely on more detailed drug labeling, multidisciplinary consultation and specialized resources such as:
- MotherToBaby (teratology information service),
- LactMed (lactation pharmacology database),
- ACOG guidelines,
- primary literature, and
- specialist pharmacy consultation.
Evidence-based interpretation rather than alphabetical classification is critical, as pregnant patients may receive misleading or overly cautious recommendations when older systems are applied uncritically.
Systemic corticosteroids in pregnancy
Systemic corticosteroids remain foundational in managing moderate-to-severe uveitis, including in pregnancy. The guiding principle is clear: If steroids are clinically necessary to preserve vision or control dangerous inflammation, they shouldn’t be withheld.2-5
There are a few key considerations to mention:
- First trimester risks: Some data suggest a small increase in the risk of orofacial clefts, though evidence is mixed and the absolute risk is low.
- Maternal side effects: Hyperglycemia is common but manageable through routine prenatal care.
- Short courses vs. chronic use: Prolonged high-dose steroid therapy is undesirable in all patients, including pregnant individuals, and warrants consideration of steroid-sparing agents.
Ultimately, uncontrolled inflammation poses a far greater risk than judicious corticosteroid use.
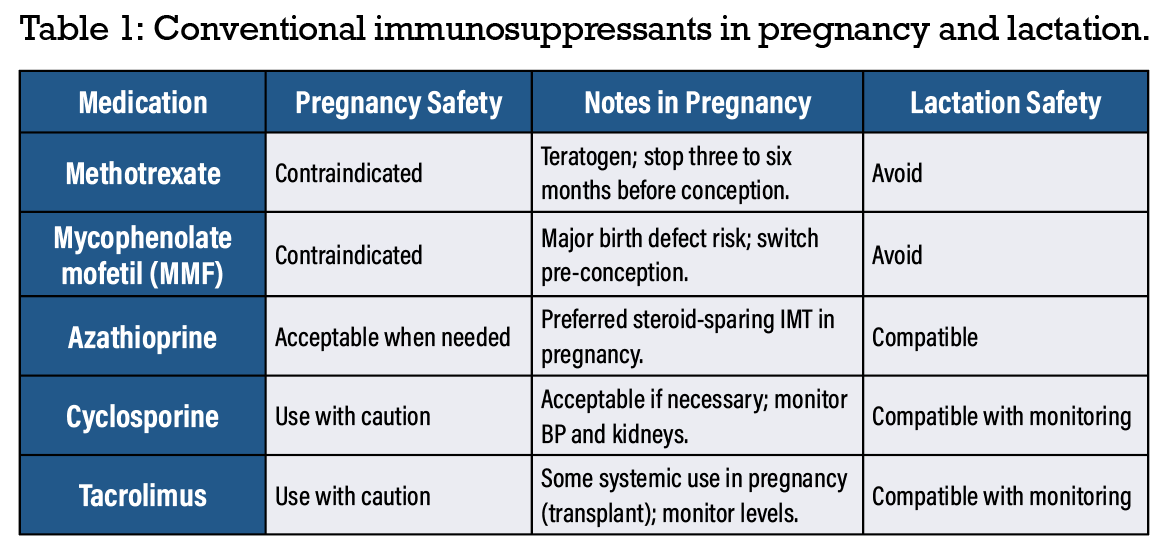 |
Antimetabolites: Contraindicated vs. acceptable options
Steroid-sparing immunomodulatory therapy is frequently necessary in chronic uveitis and requires careful reproductive planning.
The following agents are teratogenic and shouldn’t be used in pregnancy:
- Methotrexate
- Mycophenolate mofetil
Both require washout periods prior to conception. Typical recommendations include:
- Methotrexate: three to four months (some clinicians extend to six months)
- MMF: Similar guidance; transition to safer therapy as early as possible.
Azathioprine is widely considered one of the safest systemic immunosuppressants for use in pregnancy when required. While some studies associate it with low birth weight or preterm birth, these outcomes often reflect underlying disease severity rather than drug effect. For patients requiring systemic IMT, azathioprine is a strong first-line choice. Use and considerations of conventional immunosuppressives for uveitis in pregnancy is summarized in Table 1.1-8
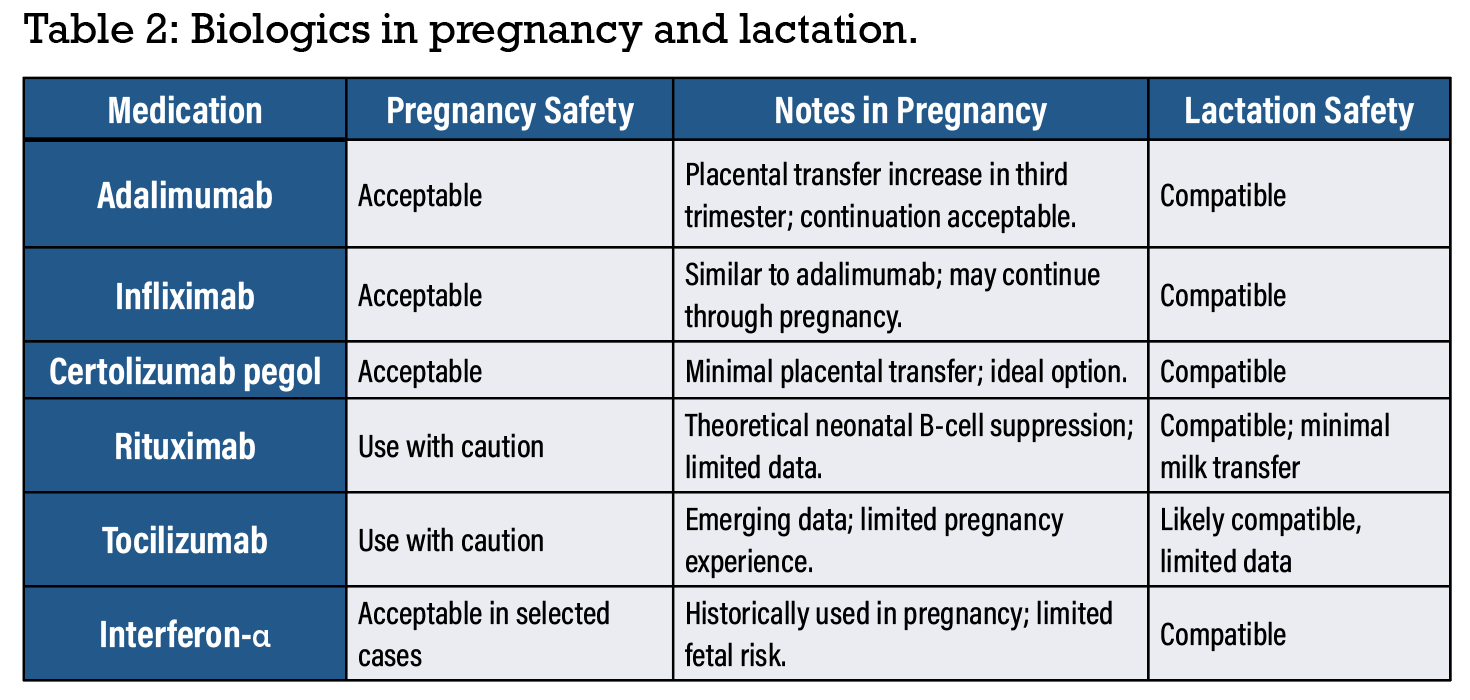
Biologic therapy during pregnancy
Biologic agents, particularly TNF-α inhibitors, play an increasingly prominent role in managing refractory uveitis. Their safety in pregnancy continues to be clarified, with growing data suggesting a more permissive approach.5,6
• TNF-α Inhibitors. Traditional practice has been to discontinue TNF inhibitors at the start of the third trimester due to placental transfer of IgG-based therapies. However, updated evidence suggests TNF inhibitors can be continued throughout pregnancy without increasing the infant’s risk of severe infections. Continuation may reduce postpartum flare and maintain better maternal disease control. Certolizumab is a uniquely attractive option during pregnancy because it lacks an Fc region, resulting in minimal placental transfer. It’s often preferred by patients concerned about fetal exposure5 and can replace other anti-TNF agents before conception if desired, although switching isn’t mandatory when disease control is excellent.
• Rituximab. Data is limited but reassuring with the major concern being theoretical neonatal B-cell depletion.
• Tocilizumab. Early data suggest limited placental transfer and cautious use with MFM collaboration is advised.
Biologic therapy selection should incorporate disease severity, response history, patient values and obstetric consultation. Considerations for use of biologics for uveitis in pregnancy is summarized in Table 2.5,6
 |
Local therapy during pregnancy
Local corticosteroid therapy—topical, periocular or intravitreal—is often preferred for unilateral or moderate inflammation, especially when systemic medication exposure is undesirable. Local therapy offers advantages such as minimal systemic absorption, rapid control of inflammation and avoidance of systemic teratogenic risk.
However, steroid-induced intraocular pressure elevation remains a concern, especially in patients known to be “steroid responders.” Still, during pregnancy—when timelines are finite—clinicians typically favor local therapy over systemic treatment for unilateral disease.
Systemic therapy for bilateral or severe disease
When uveitis is bilateral, vision-threatening or rapidly progressive, systemic therapy is necessary. High-dose systemic corticosteroids are an appropriate initial therapy even in pregnancy, followed by a slow taper. If high doses can’t be safely maintained, azathioprine or a biologic therapy may be added.
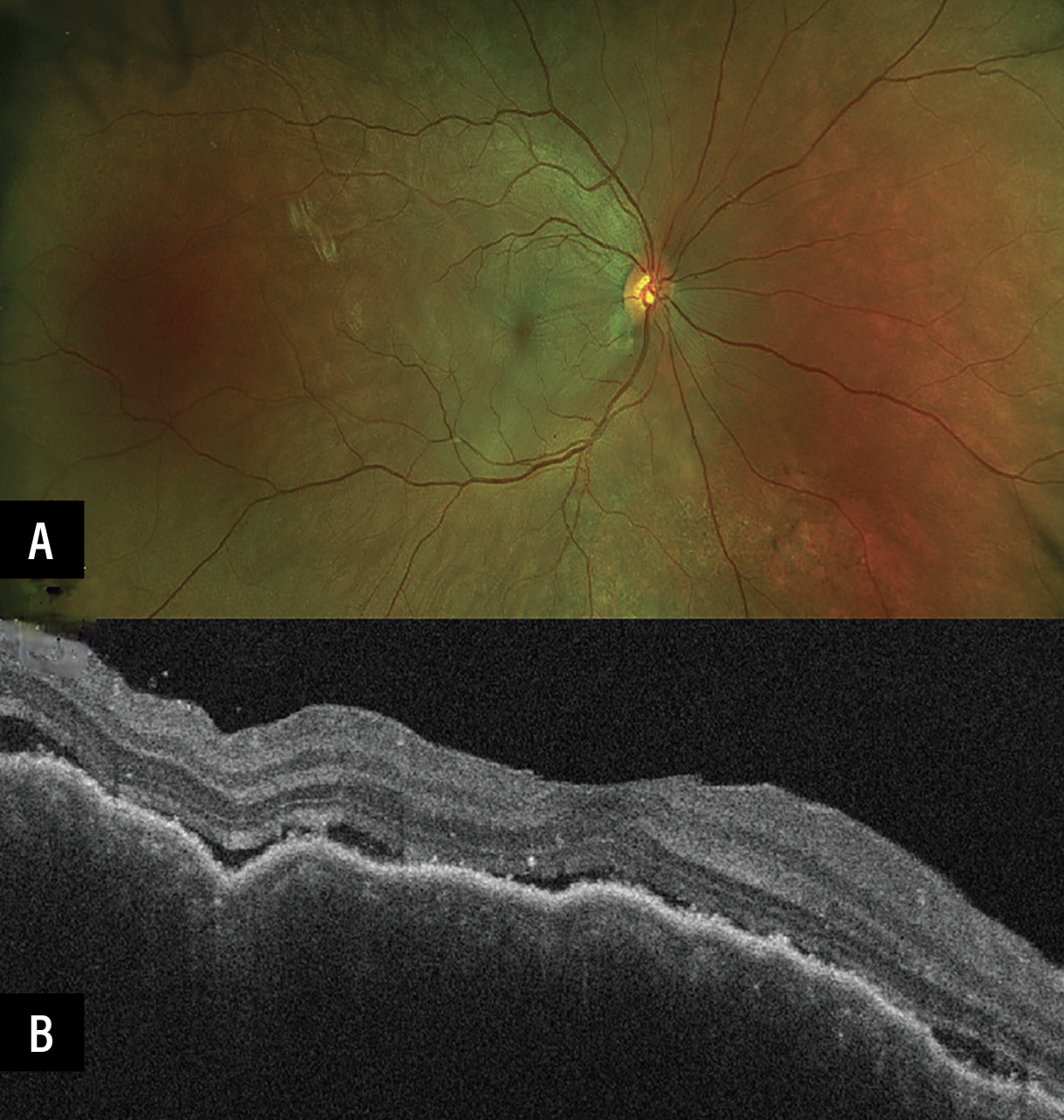 |
| A 32-year-old patient presented during the first trimester of pregnancy with bilateral serous retinal detachment (A) secondary to acute Vogt-Koyanagi-Harada disease. OCT revealed choroidal thickening and subretinal fluid (B). She required high-dose oral steroids as well as initiation of adalimumab during the pregnancy to control the inflammation. |
The decision to introduce a TNF inhibitor early is influenced by these factors:
- Trimester of pregnancy,
- Anticipated steroid taper response,
- Disease chronicity and severity, and
- Potential for postpartum exacerbation.
Avoiding undertreatment is essential. Irreversible damage from poorly controlled inflammation carries lifelong consequences.
Anti-VEGF therapy during pregnancy
Anti-VEGF injections pose a unique challenge during pregnancy, particularly in managing choroidal neovascular membranes. Systemic absorption of intravitreal agents is low but not negligible, and VEGF plays a role in placental function.9
Important guidance:
- If CNVM is active, treatment shouldn’t be delayed. Vision loss from untreated CNVM is often permanent.
- The first trimester requires the most caution due to baseline miscarriage risk and organogenesis.
- When anti-VEGF therapy is necessary, ranibizumab may be preferred because it results in the lowest systemic VEGF suppression.
- Local steroid therapy may be attempted for inflammatory lesions but is generally insufficient for a true CNVM.
In all cases, shared decision-making is crucial, balancing fetal risk perception with the danger of permanent maternal vision loss.
Monitoring pregnant patients on immunosuppression
Pregnant patients on IMT generally don’t require specialized maternal monitoring solely due to immunosuppression. Standard prenatal care applies with a few adjustments:
- Gestational diabetes screening earlier than usual for chronic steroid users.
- Close coordination with pediatrics for infants exposed to biologics, as timing of live vaccines may need modification.
- Maintenance of routine maternal immunizations: influenza; Tdap; and COVID boosters.
Collaboration with MFM specialists improves patient confidence and ensures comprehensive care.
Postpartum flares and breastfeeding considerations
The postpartum period, sometimes called the “fourth trimester,” is a high-risk window for uveitis flare due to immune rebound. Management goals include rapid control of inflammation and safe reinstitution of pre-pregnancy therapy.
For mothers who choose to breastfeed, the general principles include:
- Corticosteroids: Safe during lactation.
- Biologics: Transfer into breast milk occurs but absorption is extremely low due to GI breakdown; generally considered safe.
- Methotrexate: Contraindicated; shouldn’t be used during breastfeeding.
- Intravitreal anti-VEGF: Very low systemic levels; breastfeeding likely safe, though some patients may prefer “pump and dump” after injections for reassurance.
Conclusion
Management of uveitis in pregnancy requires an individualized, multidisciplinary approach that balances maternal visual function and fetal safety. Most patients experience a natural improvement in inflammation during mid-to-late pregnancy, yet postpartum flares are common and require vigilance. Safe and effective treatment options exist across all trimesters when guided by disease severity, pharmacologic understanding and shared decision-making.
By integrating obstetric input, proactively planning medication transitions for reproductive-age patients and applying thoughtful treatment strategies—ranging from local therapy to biologics—clinicians can successfully navigate uveitis management through pregnancy, delivery and the postpartum period while preserving maternal vision and supporting healthy pregnancy outcomes. RS
REFERENCES
1. Davis JL. Uveitis in pregnancy. J Ophthalmic Inflamm Infect 2013;3:1:49.
2. Kump LI, Cervantes-Castañeda RA, Androudi SN, Foster CS. Uveitis in pregnancy. Ocul Immunol Inflamm 2005;13:4:235-244.
3. Makri N, Ioannou K, Papadaki S, et al. Management of noninfectious uveitis during pregnancy. Clin Exp Rheumatol 2014;32:4:84:S164-S170.
4. Vinet É, Pineau CA, Clarke AE, et al. Inflammatory disease activity in pregnant women with autoimmune diseases. Rheumatology (Oxford) 2014;53:2:206-211.
5. Clowse MEB, Förger F, Hwang C, et al. Minimal to no transfer of certolizumab pegol into breast milk: Results from a prospective, postmarketing, multicentre, pharmacokinetic study. Ann Rheum Dis 2017;76:11:1890-1896.
6. Mahadevan U, Wolf DC, Dubinsky M, et al. Placental transfer of anti–tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol 2013;11:3:286-292.
7. Sadaka A, Rychwalski P, Barakat MR. Ocular manifestations of pregnancy. Middle East Afr J Ophthalmol 2016;23:2:109-114.
8. Sammaritano LR, Bermas BL, Chakravarty EE, et al. 2020 American College of Rheumatology guideline for the management of reproductive health in rheumatic and musculoskeletal diseases. Arthritis Care Res (Hoboken) 2020;72:4:461-488.
9. Gomez-Ledesma I, Valverde-Merino M, Eguizabal E, et al. Safety of intravitreal anti-VEGF therapy in pregnant women: C ase series and systematic review. Eur J Ophthalmol 2021;31:6:2960-2967.