|
|
Bios Ms. Pattathil attends Queen’s University School of Medicine in Kingston, Canada. |
As is well-known, proliferative vitreoretinopathy is a major complication that can occur following retinal detachment repair, characterized by the proliferative growth of contractile cellular membranes on the retina and in the vitreous.1 The fibrotic membranes that grow in PVR are predominantly derived from the retinal pigment epithelium, which undergo a series of inflammatory and myofibroblastic changes within the inner and outer retinal surfaces and hyaloid.2 The identification of molecular biomarkers implicated in these dysregulated inflammatory and fibrotic etiological processes could be predictive for the development of PVR, which has the potential to help stratify patients by risk and allow for targeted action earlier in the disease process.2,3
Factors involved in the stages of post-RD PVR development
PVR goes through five distinct phases in its development cycle:
1) Ischemic phase. Ischemia in the outer retina leads to diffusion of hypoxic products within the retinal space, triggering breakdown of the blood-retinal barrier within the inner retina. Within three days after RD, approximately 20 percent of photoreceptors die via necrosis, caspase-dependent apoptosis or necroptosis. These cell death pathways are mediated by receptor interacting protein kinase (RIPK1 and RIPK3). Continued ischemia triggers the upregulation of angiogenesis, fibrogenesis, glial proliferation, and the release of further growth factors and cytokines.2,4
2) Inflammatory phase. The next phase of PVR development is initiated by the release of serum factors such as thrombin into the vitreous. Macrophages secrete growth factors, specifically fibroblast growth factor and transforming growth factor-beta that stimulate the proliferation of fibroblast-like cells within growing membranes. T-helper cells are also involved in this process, as they release various anti- and pro-fibrogenic cytokines, including interleukin-10, antifibrotic interferon-gamma, FGF, TGFβ, platelet-derived growth factor (PDGF) and vascular endothelial growth factor (VEGF).2–4
3) Retinal apoptotic phase. Apoptosis is a cell loss process mediated through intrinsic and extrinsic signaling pathways that are triggered by intracellular death receptors, with considerable shared pathways with the pathogenesis of PVR. Pro-apoptotic factors are dysregulated in PVR, specifically Fas and tumor necrosis factor. Fas ligand receptor binding triggers apoptosis in proliferating RPE cells. Therefore, the FasL/Fas system, when in dysregulation, may contribute to PVR through the defective removal of excess RPE cells. TGFβ is a factor also released during this process, which contributes to the blocking of T-cell-mediated apoptosis and upregulates the proliferation of RPE cells.5–7
4) Cell migratory and proliferation phase. Epiretinal membranes consist of a collagenous core, transformed and untransformed RPE cells, Muller glia, T-lymphocytes, macrophages, astrocytes and microglia. Post-RD, the initiation of PVR begins with the activation of RPE cells and their epithelial-mesenchymal transition (EMT), leading to further proliferation, the formation of cell groups that migrate into the vitreous space, and the adoption of an extracellular matrix (ECM) and profibrotic secretory phenotype. Fibronectin initiates the production of further fibronectin, thrombospondin and other proteoglycans, while also inducing the deposition of collagen from fibroblasts. Muller glia also proliferate and release ECM and profibrotic and inflammatory mediators.2,7
5) Scar contraction phase. Within PVR membranes, transformed cells can differentiate into myofibroblasts through stimulation from IL-1. Annexin A2 mediates contractile ability in myofibroblasts, which releases further RPE cells into the vitreous space and worsens retinal detachment.2,3,8
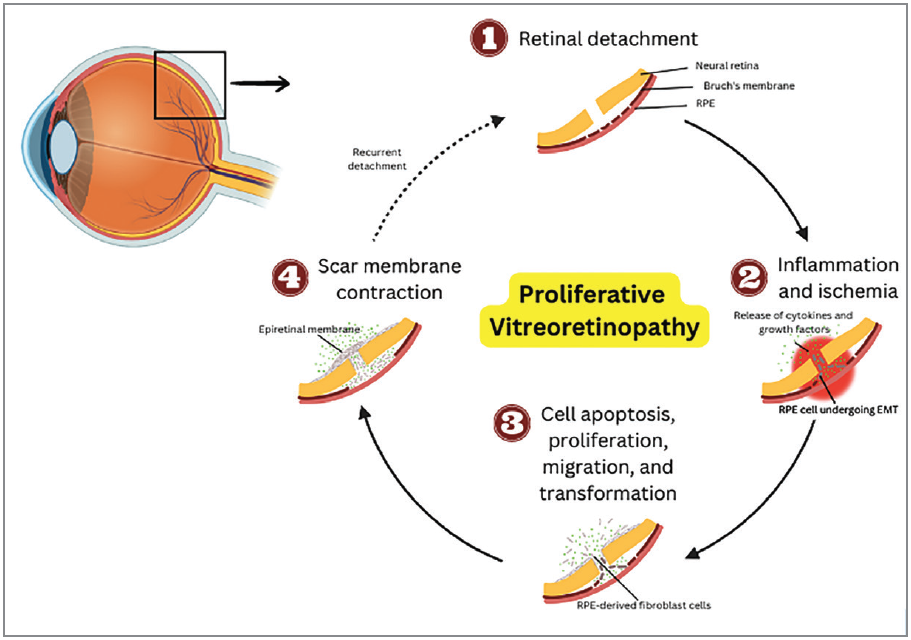 |
| Summary diagram of pathophysiology of proliferative vitreoretinopathy. |
Potential biomarkers for prediction of PVR after RD
In the literature, the following cytokines and growth factors have been identified within subretinal fluid to be potentially predictive of PVR development:
• Interleukins 1a, 2, 3, 6, 11, 15, 18;
• Chemokine Ligands 2, 3, 11, 17, 18,
19, 22;
• CXC Ligands 8, 9, 10;
• CTSS;
• ADIPOQ;
• Leptin;
• ICAM-1;
• VCAM-1;
• PDGF;
• VEGF;
• Fas;
• FasL; and
• TIMP-1.2,7
There are also several cytokines and growth factors that have been identified within vitreous samples to be potentially predictive of PVR development:
• IL-6;
• TGFB2;
• FGF-2;
• Tot Prot;
• MMP-2;
• MMP-9;
• Cont Stim Fac;
• Decorin; and
• miR-21.9,10
Challenges in the validation of molecular biomarkers
Predictive molecular biomarkers are substances found in a target tissue that can help indicate the risk of developing a specific pathology. Evaluation of biomarkers relies on the reliability of the sampling technique as obtaining consistent samples of subretinal fluid and vitreous fluid can be challenging and therefore lead to highly variable values. Serum samples can be more reliable but may have lower biomarker levels compared to levels in local retinal production. Additionally, surgical techniques for RD repair can vary, which could potentially influence the rates of PVR between various surgical approaches.2,11
Therefore, future biomarker studies should consider detailing the specific surgical approaches taken by their sample or include a range of surgeons and techniques in their analyses. In patients with RD, it’s challenging to identify biomarkers with high positive predictive value for PVR development because most molecules are also present in RD cases irrespective of PVR development. Therefore, future research should focus on combining specific biomarkers and expanding screening methods to include other potential factors in order to improve the prediction methods for PVR.3,12
Conclusion
In summary, PVR is the leading cause of failure following surgery for repair of RD. The retinal fibrosis of PVR is triggered by an abnormally regulated array of inflammatory markers, cytokines and growth factors which produce an aberrant inflammatory response at the site of retinal detachment or tear. The process is initiated by RPE-derived fibroblasts that undergo epithelial-mesenchymal transition and start depositing extracellular matrix components and collagen on the retinal surfaces.2,9,11
Biomarker profiling holds promise in predicting the development of PVR after surgery, which could help guide management and identify patients earlier who are at risk and may benefit from prophylactic therapy. Current evidence suggests that there are various inflammatory and fibrogenic factors that are associated with RD and can also contribute to the pathogenesis of PVR. Factors with persistent presence into the PVR state may serve as prognostic biomarkers and also be useful as potential targets for anti-PVR treatments.1,6,10 Individual factors have limited positive predictive value and most have significant overlap between their levels in patients who did and did not go on to develop PVR, which limits utility as a biomarker in isolation. Therefore, the most promising approach is combining multiple clinical and laboratory biomarkers to improve the sensitivity and specificity of PVR prediction.2,5,7,8 RS
References
1. Mudhar HS. A brief review of the histopathology of proliferative vitreoretinopathy (PVR). Eye 2020;34:246–50.
2. Chaudhary R, Scott RAH, Wallace G, Berry M, Logan A, Blanch RJ. Inflammatory and fibrogenic factors in proliferative vitreoretinopathy development. Transl Vis Sci Technol 2020;9:3:23.
3. Charteris DG. Proliferative vitreoretinopathy: revised concepts of pathogenesis and adjunctive treatment. Eye 2020;34:241–5.
4. Dai Y, Dai C, Sun T. Inflammatory mediators of proliferative vitreoretinopathy: Hypothesis and review. Int Ophthalmol 2020;40:1587–601.
5. Wong CW, Cheung N, Ho C, Barathi V, Storm G, Wong TT. Characterisation of the inflammatory cytokine and growth factor profile in a rabbit model of proliferative vitreoretinopathy. Sci Rep 2019;9:15419.
6. El Ghrably I, Powe DG, Orr G, Fischer D, McIntosh R, Dua HS, et al. Apoptosis in proliferative vitreoretinopathy. Investigative Ophthalmology & Visual Science 2004;45:1473–9.
7. Idrees S, Sridhar J, Kuriyan AE. Proliferative vitreoretinopathy: A review. Int Ophthalmol Clin 2019;59:221–40.
8. Moysidis SN, Thanos A, Vavvas DG. Mechanisms of inflammation in proliferative vitreoretinopathy: From bench to bedside. Mediators of Inflammation 2012;2012:e815937.
9. Yu J, Liu F, Cui S-J, Liu Y, Song Z-Y, Cao H, et al. Vitreous proteomic analysis of proliferative vitreoretinopathy. PROTEOMICS 2008;8:3667–78.
10. Ni Y, Qin Y, Huang Z, Liu F, Zhang S, Zhang Z. Distinct serum and vitreous inflammation-related factor profiles in patients with proliferative vitreoretinopathy. Adv Ther 2020;37:2550–9.
11. Shahlaee A, Woeller CF, Philp NJ, Kuriyan AE. Translational and clinical advancements in management of proliferative vitreoretinopathy. Current Opinion in Ophthalmology 2022;33:219–27.
12. Ricker LJAG, Kessels AGH, de Jager W, Hendrikse F, Kijlstra A, la Heij EC. Prediction of proliferative vitreoretinopathy after retinal detachment surgery: Potential of biomarker profiling. American Journal of Ophthalmology 2012;154:347-354.e2.