Take-home points
|
 |
Bios Dr. Zhou is a PGY-3 ophthalmology resident at the Mayo Clinic in Rochester, Minnesota. |
Glucagon-like peptide 1 receptor agonists are paradigm-shifting medications, helping many patients manage their diabetes, aiding with weight loss and reducing risk of serious health complications. Even with their significant potential benefits, however, GLP-1 RAs come with safety warnings for patients to speak with their doctor before starting treatment if they have a history of diabetic retinopathy. Patients and colleagues in other specialties are going to seek the input of retina specialists regarding the use of GLP-1 RAs. Therefore, it’s imperative to understand what the data tells us about GLP-1 RAs and diabetic retinopathy. Here we’ll outline what you need to know to aid patients and your colleagues when they come to you for answers.
The rise of GLP-1 RAs
As a retinal specialist, many of your patients are already using GLP-1 RAs and many more will receive new prescriptions in the near future. Survey data from the National Center for Health Statistics published in 2025 show that more than a quarter (26.5 percent) of adults diagnosed with diabetes are using GLP-1 RAs.1 In addition to the growth in usage among patients with diabetes, GLP-1 RA prescriptions are rising rapidly in the broader U.S. population, with the share of GLP-1 RA fills for non-diabetes-related conditions nearly doubling from 7.4 percent in 2019 to 14.3 percent in 2022.2
There are six FDA-approved GLP-1 RAs on the market, including several that are widely recognized in the popular media: exenatide (Byetta, Bydureon); liraglutide (Victoza, Saxenda); dulaglutide (Trulicity); lixisenatide (Adlyxin); semaglutide (Ozempic, Rybelsus, Wegovy); and tirzepatide (Mounjaro, Zepbound, which works as a dual GLP-1/GIP [glucose-dependent insulinotropic polypeptide] agonist). Recently, an oral formulation of Wegovy and a new oral GLP-1, Foundayo (Lilly), were approved, which could increase adoption rates of these drugs even further.
These agents mimic GLP-1, an endogenous incretin hormone released in response to eating. In doing so, GLP-1 RAs stimulate insulin release while suppressing glucagon secretion and slowing gastric emptying, ultimately reducing appetite.3 But the effects of GLP-1 RAs are even more sweeping. GLP-1 receptors are present in many tissues ranging from the brain to the heart to the retinal pigment epithelium.4,5 In the retina, pre-clinical studies have revealed potentially anti-inflammatory and neuroprotective effects independent of glucose-lowering effects.6
Importantly, these are life-saving therapies, independent of any impact they may have on the eye. A recent meta-analysis of 11 randomized controlled trials demonstrated a 13-percent relative risk reduction for all-cause death, along with significant risk reductions across multiple outcomes, including 16 percent for renal failure, 13 percent for congestive heart failure hospitalizations, 14 percent for major adverse cardiovascular events, 12 percent for stroke, 13 percent for non-fatal myocardial infarction and 14 percent for cardiovascular death.7 Recognition of the role GLP-1 RAs play in systemic health has been reflected in clinical guidelines, with GLP-1 RAs recommended as first-line in the pharmacologic management of type 2 diabetes by the American Diabetes Association for patients with various systemic co-morbidities.8
 |
GLP-1 RAs and DR: What the data show
The CDC’s Vision and Eye Health Surveillance System estimated that in 2021, there were 9.6 million people (26 percent of those with diabetes) living with diabetic retinopathy in the United States and among them, 1.84 million people (5 percent) living with vision-threatening diabetic retinopathy.9 Many of these patients are currently using or will eventually use GLP-1 RAs. Pre-clinical studies have demonstrated mixed findings, including potential protective effects of GLP-1 RAs in the context of diabetic retinopathy, with GLP-1 receptors found in the RPE serving a neuroprotective role in conditions of high glucose.6
Yet in 2016, data from the SUSTAIN-6 trial comparing semaglutide to placebo over two years showed a possible increased risk of complications of diabetic retinopathy, including vitreous hemorrhage; the need for intravitreal injections and laser; and blindness (hazard ratio, 1.76; 95% CI, 1.11 to 2.78; p=0.02).10 However, in the SUSTAIN-6 trial, there was also a significant drop in hemoglobin A1c (>1 percent) among participants receiving semaglutide during the first 16 weeks of GLP-1 RA therapy. The Diabetes Control and Complications Trial research group previously observed that early worsening of diabetic retinopathy can occur with intensive glycemic control, though effective glycemic control ultimately lowers retinopathy risk over the long-term.11,12 The risk of complications in SUSTAIN-6 was highest among patients with pre-existing diabetic retinopathy and rapid HbA1c reduction, suggesting that the findings reflect the early worsening of diabetic retinopathy that can occur with any method of lowering glucose, rather than direct retinal toxicity from GLP-1 RAs.
Other trials of GLP-1 RAs have also not yet provided a clear answer. Most of the studies were designed to evaluate cardiovascular or renal outcomes, with diabetic retinopathy data collected through inconsistent and variably rigorous adverse events reporting. In 2004, researchers conducted a meta-analysis of 61 randomized controlled trials evaluating GLP-1 RAs, sodium-glucose co-transporter-2 inhibitors (SGLT2i), dipeptidyl peptidase 4 inhibitors (DPP-4i) and didn’t find an increased risk of diabetic retinopathy events with GLP-1 RAs or any of the other classes of antihyperglycemic medications.13
Another study examined 93 trials and found an increased risk of diabetic retinopathy and retinal adverse effects with GLP-1 RA use, though this effect appeared driven by a single agent, albiglutide, which is no longer available.14 Both studies were limited by relatively short follow-up among the included trials, narrow systemic inclusion criteria, limited information on baseline diabetic retinopathy status and, most important, inadequate and non-rigorous methodologies for diabetic retinopathy evaluation.13,14
Similarly, analysis of data from routine clinical practice has yielded varying results. One analysis used medical and pharmacy claims data to evaluate outcomes in 371,698 patients with type 2 diabetes initiating GLP-1 RA, SGLT2i, DPP-4i and sulfonylurea class medications, with the primary outcome being time-to-treatment for DME or PDR.15 While the study showed a lower risk of sight-threatening retinopathy with SGLT2i, there was no increased risk with GLP-1 RAs compared with DPP-4i or sulfonylureas. Similarly, another study found a higher rate of progression of proliferative diabetic retinopathy as well as risk of new-onset diabetic macular edema with GLP-1 RAs compared to SGLT-2is.16 However, a separate study didn’t find a significant difference in clinical worsening of retinopathy or in need for intervention between GLP-1 RAs and SGLT-2i’s.17 Similarly, a group from Taiwan analyzed data from the Taiwan National Health
Insurance Research Database and ultimately didn’t find an increased risk of vision-threatening retinopathy with GLP-1 RA use and a possible decreased risk compared to DPP-4i.18
Just as there are differences between the antihyperglycemic agents, there are inter-agent differences between the GLP-1 RAs, with established differences in their glucose-lowering, lipid-lowering and weight loss effects. However, a study that performed three-way (exenatide vs. dulaglutide vs. liraglutide) and two-way (semaglutide vs. dulaglutide) comparisons among 125,336 adults with type 2 diabetes initiating a GLP-1 RA and found no difference in the rate of sight-threatening diabetic retinopathy between the different GLP-1 RA agents.19 One report compared semaglutide with other GLP-1 RAs (dulaglutide, exenatide) and with non-GLP-1 RA antihyperglycemics (SGLT-2i, DPP4i, sulfonylurea).20 The researchers didn’t find an increased risk for proliferative diabetic retinopathy or for diabetic retinopathy/diabetic macular edema requiring treatment with semaglutide compared to the other GLP-1 RAs or to the non-GLP-1 RA antihyperglycemic medications.
While the studies published to date have limitations, there are several ongoing investigations, including the pivotal FOCUS trial, conducted as a post-authorization safety commitment to the European Medicines Agency.21 The study has recruited 1,500 patients at 176 sites worldwide and compares semaglutide versus placebo in patients with type 2 diabetes. The primary outcome will be the percentage of participants with ≥3 step Early Treatment Diabetic Retinopathy Study (ETDRS) subject-level progression at five years, as well as secondary outcomes including 17 ocular and four systemic outcomes. The study will conclude in 2027 with results anticipated in early 2028.
In the meantime, retina specialists can counsel patients and the interdisciplinary care team that the data published to date haven’t demonstrated definitive evidence of harm or an increased risk of diabetic retinopathy progression or vision-threatening adverse events with GLP-1 RA initiation. The principles of managing diabetic retinopathy remain unchanged in patients receiving a GLP-1 RA.
What to watch for and how to manage
If a patient presents with suspected issues with a GLP-1 RA drug, here’s how to approach them:
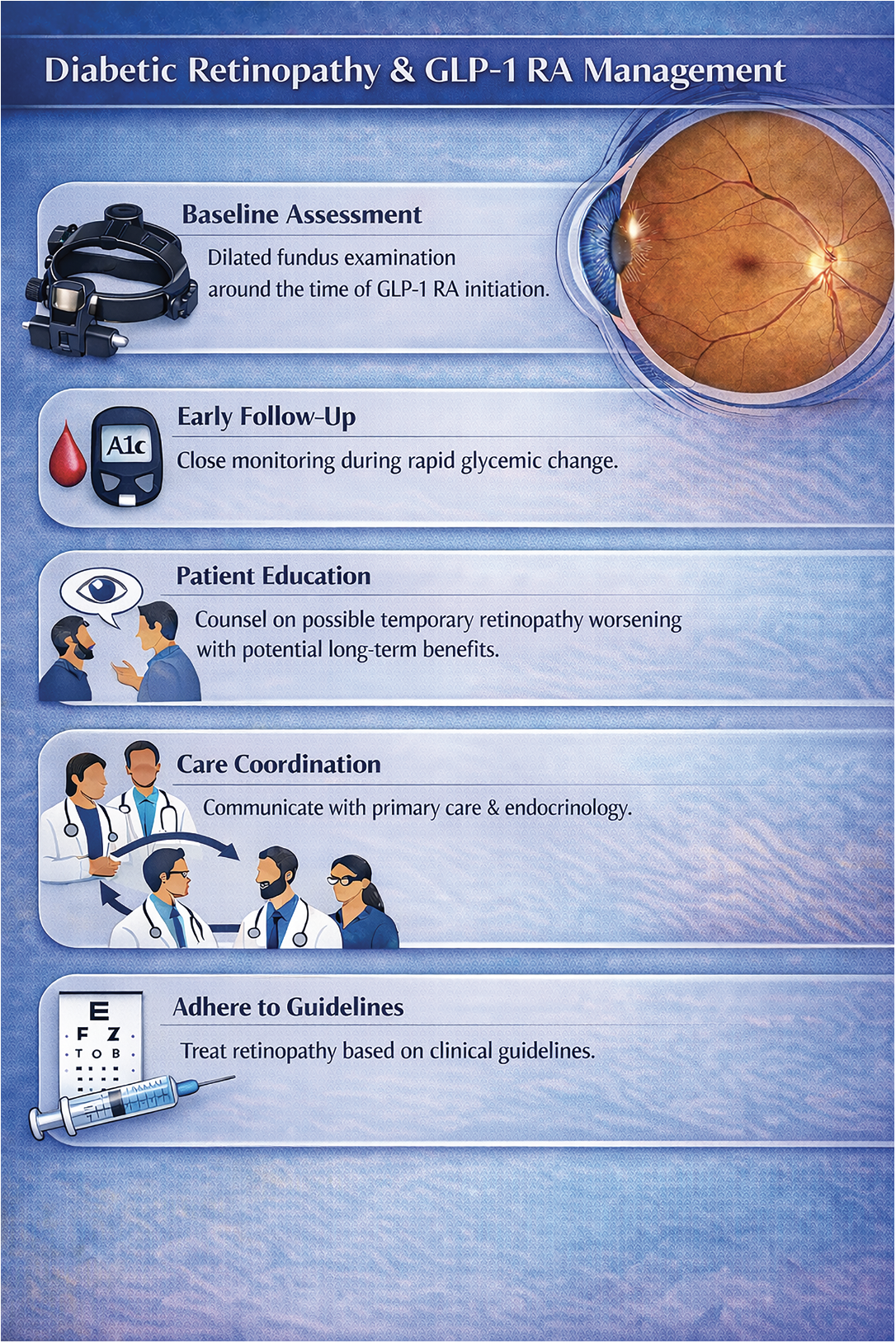
• Baseline assessment. Baseline evaluation with dilated fundus examination prior to or shortly after GLP-1 RA initiation is recommended, particularly in patients with more advanced diabetic retinopathy. This helps clinicians determine how closely to follow patients after GLP-1 RA initiation as they may be at increased short-term risk.
• Early follow-up during glycemic change. Patients with diabetes who are initiating GLP-1 RAs—especially those with elevated baseline hemoglobin A1c or anticipated rapid reduction—may benefit from being monitored more closely than suggested by the American Academy of Ophthalmology preferred practice pattern, which is based on the current level of retinopathy. This mirrors management strategies used in other scenarios with significant anticipated glycemic change, such as pregnancy, intensive insulin therapy or bariatric surgery.
• Patient counseling and education. Patients should be counseled ahead of time that transient worsening of diabetic retinopathy or macular edema can happen whenever there is rapid glucose improvement, but that there are significant long-term benefits for diabetic retinopathy. Temporary worsening after GLP-1 RA initiation doesn’t necessarily imply that GLP-1 RAs are harmful to the eye or should be discontinued. Setting expectations eases anxiety, improves adherence and reduces unnecessary discontinuation of therapy.
• Coordination with prescribing clinicians. Communication with the patient’s care team, including primary care providers and endocrinologists, is essential. Management of diabetic retinopathy requires concurrent management of diabetes and other systemic parameters.
• Treatment of diabetic retinopathy according to established clinical guidelines. Management of diabetic retinopathy should follow established clinical guidelines and practice patterns, including anti-VEGF therapy, laser or surgery as indicated.
Conclusions
Ultimately, while further investigation will help clarify the risks and benefits of GLP-1 RAs with respect to diabetic retinopathy and vision-threatening complications, it’s clear that GLP-1 RAs confer proven mortality and cardiovascular benefit. There’s currently no convincing evidence for avoiding or delaying GLP-1 RAs out of concern for diabetic retinopathy progression. There is, however, evidence to support considering closer follow-up when initiating a GLP-1 RA, particularly if rapid improvement in glycemic control is anticipated, following a baseline dilated fundus examination performed around the time of therapy initiation. Patients should be counseled that close follow-up is recommended and that there can be temporary worsening of diabetic retinopathy and diabetic macular edema with rapid intensive glycemic control. As GLP-1 RAs become increasingly prevalent, retina specialists play a critical role in ensuring that patients receive the systemic benefits of these agents while minimizing ocular risk through surveillance, education and collaboration. RS
REFERENCES
1. Vahratian A, Warren A. GLP-1 injectable use among adults with diagnosed diabetes: United States, 2024. NCHS Data Brief 2024;537:1-8.
2. Hegland TA, Fang Z, Bucher K. GLP-1 Medication use for type 2 diabetes has soared. JAMA 2024;332:12:952-953.
3. Doyle ME, Egan JM. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol Ther 2007;113:3:546-93.
4. Zhao X, Wang M, Wen Z, et al. GLP-1 receptor agonists: Beyond their pancreatic effects. Front Endocrinol (Lausanne) 2021;12:721135.
5. Puddu A, Sanguineti R, Montecucco F, Viviani GL. Retinal pigment epithelial cells express a functional receptor for glucagon-like peptide-1 (GLP-1). Mediators Inflamm 2013;2013:975032.
6. Zhou HR, Ma XF, Lin WJ, et al. Neuroprotective role of GLP-1 analog for retinal ganglion cells via PINK1/Parkin-Mediated Mitophagy in diabetic retinopathy. Front Pharmacol 2020;11:589114.
7. Badve SV, Bilal A, Lee MMY, et al. Effects of GLP-1 receptor agonists on kidney and cardiovascular disease outcomes: A meta-analysis of randomised controlled trials. Lancet Diabetes Endocrinol 2025;13:1:15-28.
8. American Diabetes Association professional practice committee for diabetes. pharmacologic approaches to glycemic treatment: Standards of care in diabetes—2026. Diabetes Care 2026;49:S183-S215.
9. Lundeen EA, Burke-Conte Z, Rein DB, et al. Prevalence of diabetic retinopathy in the US in 2021. JAMA Ophthalmol 2023;141:8:747-754.
10. Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with Type 2 Diabetes. N Engl J Med 2016;375:19:1834-1844.
11. Diabetes C, Complications Trial Research G, Nathan DM, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:14:977-86.
12. Early worsening of diabetic retinopathy in the Diabetes Control and Complications Trial. Arch Ophthalmol 1998;116:7:874-86.
13. Malyszczak A, Przezdziecka-Dolyk J, Szydelko-Pasko U, Misiuk-Hojlo M. Novel antidiabetic drugs and the risk of diabetic retinopathy: A systematic review and meta-analysis of randomized controlled trials. J Clin Med 2024;13:6.
14. Kapoor I, Sarvepalli SM, D’Alessio D, Grewal DS, Hadziahmetovic M. GLP-1 receptor agonists and diabetic retinopathy: A meta-analysis of randomized clinical trials. Surv Ophthalmol 2023;68:6:1071-1083.
15. Barkmeier AJ, Herrin J, Swarna KS, et al. Comparative effectiveness of glucagon-like peptide-1 receptor agonists, sodium-glucose cotransporter 2 inhibitors, dipeptidyl peptidase-4 inhibitors, and sulfonylureas for sight-threatening diabetic retinopathy. Ophthalmol Retina 2024;8:10:943-952.
16. Wai KM, Mishra K, Koo E, et al. Impact of GLP-1 agonists and SGLT-2 inhibitors on diabetic retinopathy progression: An aggregated electronic health record data study. Am J Ophthalmol 2024;265:39-47.
17. Joo JH, Sharma N, Shaia J, et al. The effect of glucagon-like peptide-1 receptor agonists on diabetic retinopathy at a tertiary care center. Ophthalmol Sci 2024;4:6:100547.
18. Yen FS, Wei JC, Shih YH, et al. Glucagon-like peptide-1 receptor agonists and risk of sight-threatening retinopathy in Taiwanese population: A propensity based cohort study. Diabetes Metab Syndr 2024;18:8:103099.
19. Barkmeier AJ, Deng Y, Swarna KS, et al. Risk of sight-threatening diabetic retinopathy with glucagon-like peptide-1 receptor agonist use in routine clinical practice: Comparative effectiveness of semaglutide, dulaglutide, liraglutide, and exenatide. Ophthalmol Retina 2025. August 6, 2025:S2468-6530(25)00355-0. Online ahead of print.
20. Cai CX, Nishimura A, Baxter S, et al. Semaglutide and diabetic retinopathy: An OHDSI network study. BMJ Open Diabetes Res Care 2025;13:6.
21. A Research Study to Look at How Semaglutide Compared to Placebo Affects Diabetic Eye Disease in People With Type 2 Diabetes (FOCUS). NCT03811561. ClinicalTrials.gov.